Abstract
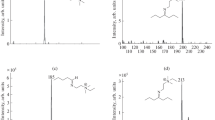
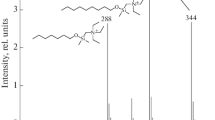
Chemical ionization (CI) mass spectra of various natural and synthetic steroids have been studied using methane, isobutane, ammonia, trideuterioammonia and hydroxy anion as reagent gases. The CI spectra of steroids give simple and well characterized ions, which provide information about molecular weight as well as functionalities in the molecules. Trideuterioammonia exchanges rapidly with active hydrogens (e.g., OH, SH, COOH, NH2) in steroid molecules in the CI reaction and thus provides a convenient means of active hydrogen determination by mass spectrometry. Application of various CI processes to the analysis of steroids and conjugates have been made. Low levels of hydroxycholesterols in biological samples and in cholesterol autoxidation products were identified by the 4 ion patterns, [M+NH4]+, [M−OH+NH3]+, [M−OH]+ and [M−H2 O−OH]+, in ammonia CI. The position of hydroxy functions in the cholesterol side chain can be identified from the methane CI of hydroxycholesterol trimethylsilyl (TMS) derivatives. Sterol carboxylic esters can be identified as the ammonium adduct ion of the intact molecule, [M+NH4]+, in ammonia CI. Isobutane and hydroxy anion CI spectra of the steroid esters give abundant ion fragments of both steroids and carboxylic acid moieties. Identification of free bile acids and steroid glycosides without derivatization is also feasible with the CI process when ammonia is used as reagent gas.
Similar content being viewed by others
References
Budzikiewicz, H., C. Djerassi and D.H. Williams, “Mass Spectrometry of Organic Compounds,” Holden-Day, San Francisco, 1967.
Zaretiskii, Z.V., “Mass Spectrometry of Steroids,” John Wiley & Sons, New York and Israel University Press, Jerusalem, 1976.
Unruh, G.V. and G. Spiteller, Tetrahedron 26:3329 (1970).
Munson, M.S.B. and F.H. Field, J. Am. Chem. Soc. 88:2621 (1966).
Field, F.H., in “Ion Molecule Reaction,” edited by J.L. Franklin, Plenum Press, New York, 1972, p. 133.
Munson, B., Anal. Chem. 49:772A (1977).
Lin, Y.Y. and L.L. Smith, Biomed. Mass Spectrom. 5:604 (1978).
Lin, Y.Y. and L.L. Smith, Biomed. Mass Spectrom. 6:15 (1979).
Roy, T.A., F.H. Field, Y.Y. Lin and L.L. Smith, Anal. Chem. 51:272 (1979).
Haney, M.A. and J.L. Franklin, J. Chem. Phys. 50:2028 (1969).
Dzic, I. and J.A. McCloskey, Org. Mass Spectrom. 6:939 (1972).
Hunt, D.F., C.N. McEwen and R.A. Upham, Tetrahedron Lett. 4439 (1971).
Hunt, D.F., C.N. McEwen and R.A. Upham, Anal. Chem. 44:1292 (1972).
Blum, W., E. Schlumpf, J.G. Liehr and W.J. Richter, Tetrahedron Lett. 565 (1976).
Zaretskii, V.I., N.S. Wulfson, V.G., Zaikin and I.V. Torger, Izv. Akad. Nauk SSSR Ser. Khim. 1294 (1978).
Grostic, M.F. and K.L. Rinehart, Jr., J. Org. Chem. 33:1740 (1968).
Gustafsson, J.A. and J. Sjoval, Eur. J. Biochem. 8:467 (1969).
Brooks, C.J.W., W. Henderson and G. Steel, Biochim. Biophys. Acta 296:431 (1973).
Gaskell, S.J., A.G. Smith and C.J.W. Brooks, Biomed. Mass Spectrom. 2:148 (1975).
Murata, T., S. Takahasi and T. Takeda, Anal. Chem. 47:577 (1975).
Szczepanik, P.A., D.L. Hachey and P.D. Klein, J. Lipid Res. 17:314 (1976).
Muschik, G.M., L.H. Wright and J.A. Schroer, Biomed. Mass Spectrom. 6:266 (1979).
Komori, T., Y. Ida, Y. Mutou, K. Miyahara, T. Nohara and T. Kawasaki, Biomed. Mass Spectrom. 2:65 (1975).
Miyazaki, H., M. Ishibaski, M. Itoh, N. Morishita, M. Sudo and T. Nambara, Biomed. Mass Spectrom. 3:55 (1976).
Speigelhalder, D., G., Röhle, L. Siekmann and H. Breuer, J. Steroid Biochem. 7:74 (1976).
Vine, J., L. Brown, J. Boutage, R. Thomas and D. Nelson, Biomed. Mass Spectrom. 6:415 (1979).
Baldwin, M.A. and F.W. McLafferty, Org. Mass Spectrom. 7:1953 (1973).
Author information
Authors and Affiliations
About this article
Cite this article
Lin, Y.Y. Identification of steroids by chemical ionization mass spectrometry. Lipids 15, 756–763 (1980). https://doi.org/10.1007/BF02534029
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02534029