Abstract
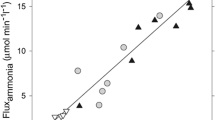
The effect of ammonia onl-glutamate (L-GLU) uptake was examined in cultured astrocytes. Acute ammonia treatment (5–10 mM) enhanced L-[3H]GLU uptake by 20–42% by increasing the Vmax; this persisted for 2 days and then started to decline. Ammonia, however, did not affect the uptake ofd-[3H]aspartate (D-ASP), a non-metabolizable analog of L-GLU, that uses the same transport carrier as L-GLU. Also, L-GLU uptake was not affected during the first 2 min of the assay. Thus, ammonia did not have an acute effect on L-GLU transport (translocation); rather, ammonia enhanced the accumulation or “trapping” of L-GLU or its by-products. Chronic ammonia treatment, on the other hand, inhibited L-GLU transport in astrocytes by ∼30–45% and this was due to a decrease in Vmax, suggesting that the number of L-GLU transporters was decreased. This inhibitory effect was observed after 1 day of treatment and persisted for at least 7 days. The inhibition of L-GLU transport was partially reversible following removal of ammonia. The effects of ammonia on L-GLU transport and uptake may explain the abnormal L-GLU neurotransmission observed in hyperammonemia/hepatic encephalopathy, and the brain swelling associated with fulminant hepatic failure.
Similar content being viewed by others
References
Mousseau, D. D., and Butterworth, R. F. 1994. Current theories on the pathogenesis of hepatic encephalopathy. Proc. Soc. Exp. Biol. Med. 206:329–344.
Rao, V. L. R., Murthy, Ch. R. K., and Butterworth, R. F. 1992. Glutamatergic synaptic dysfunction in hyperammonemic syndromes. Metab. Brain Dis. 7:1–20.
Szerb, J. C., and Butterworth, R. F. 1992. Effect of ammonium ions on synaptic transmission in the mammalian central nervous system. Prog. Neurobiol. 39:135–153.
Bosman, D. K., Deutz, N. E., de Graaf, A. A., van Eijk, H. M., Bovee, W. M., Maas, M. A., Jorning, G. G., and Chamleau, R. A. 1990. Changes in brain metabolism during hyperammonemia and acute liver failure: results of a comparative 1H-NMR spectroscopy and biochemical investigation. Hepatology 12:281–290.
Hindfelt, B., Plum, F., and Duffy, T. E., 1977. Effect of acute ammonia intoxication on cerebral metabolism in rats with portacaval shunts. J. Clin. Invest. 59:386–396.
Hamberger, A., Lindroth, B., and Nystrom, B. 1982. Regulation of glutamate biosynthesis and release in vitro by low levels of ammonium ions. Brain Res. 237:339–350.
Benjamin, A. M., Okamoto, K., and Quastel, J. H. 1978. Effects of ammonium ions on spontaneous action potentials and on contents of sodium, potassium, ammonium and chloride ions in brain in vitro. J. Neurochem. 30:131–143.
De Knegt, R. J., Schalm, S. W., Van Der Rijt, C. C. D., Fekkes, D., Dalm, E., and Hekking-Weyma, I. 1994. Extracellular brain glutamate during acute liver failure and during acute hyperammonemia simulating acute liver failure: An experimental study based on in vivo brain dialysis. J. Hepatol. 20:19–26.
Moroni, F., Lombardi, G., Moneti, G., and Cortesini, C. 1983. The release and the neosynthesis of glutamic acid are increased in experimental models of hepatic encephalopathy. J. Neurochem. 40:850–854.
Tossman, U., Delin, A., Eriksson, L. S., and Ungerstedt, U. 1987. Brain cortical amino acids measured by intracerebral dialysis in protacaval shunted rats. Neurochem. Res. 12:265–269.
Schousboe, A., Drejer, J., and Hertz, L. 1988. Uptake and release of glutamate and glutamine in neurons and astrocytes in primary cultures. Pages 21–39,in E. Kvamme, E. (ed.), Glutamine and Glutamate in Mammals, vol. II, CRC Press, Boca Raton, FL.
Danbolt, N. C., Storm-Mathisen, J., and Kanner, B. I. 1992. [Na−+K+] coupledl-glutamate transporter purified from brain is located in glia cell processes. Neuroscience 51:295–310.
Lehre, K. P., Levy, L. M., Ottersen, O. P., Storm-Mathisen, J., and Danbolt, N. C. 1995. Differential expression of two glial glutamate transporters in the rat brain: quantitative and immunocytochemical observations. J. Neurosci. 15:1835–1853.
Levy, L. M., Lehre, K. P., Rolstad, B., and Danbolt, N. C. 1993. A monoclonal antibody raised against an [Na++K+] coupledl-glutamate transporter purified from rat brain confirms glial cell localization. FEBS Lett. 317:79–84.
Rothstein, J. D., Martin, L., Levey, A. I., Dykes-Hoberg, M., Jin, L., Wu, D., Nash, N., and Kuncl, R. W. 1994. Localization of neuronal and glial glutamate transporters. Neuron 13:713–725.
Torp, R., Danbolt, N. C., Babaie, E., Bjoras, M., Seeberg, E., Storm-Mathisen, J., and Ottersen O. P. 1994. Differential expression of two glial glutamate transporters in the rat brain: an in situ hybridization study. Eur. J. Neurosci. 6:936–942.
Kondo, K., Hashimoto, H., Kitanaka, J., Sawada, M., Suzumura, A., Marunouchi, T., and Baba, A. 1995. Expression of glutamate transporters in cultured glial cells. Neurosci. Lett. 188:140–142.
Norenberg, M. D. 1981. The astrocyte in liver disease. Pages 303–352,in Fedoroff, S., and Hertz, L. (eds.), Advances in Cellular neurobiology, vol. 2, Academic Press, New York, NY.
Norenberg, M. D. 1987. The role of astrocytes in hepatic encephalopathy. Neurochem. Pathol. 6:13–34.
Bender, A.S., Hazell, A. S., and Norenberg, M. D. 1995. Acute and chronic effects of ammonia onl-glutamate uptake in cultured astrocytes. (Abstr.) J. Neurochem. 64 (Suppl.):S66C.
Norenberg, M. D., Mozes, L. W., Papendick, R. E., and Norenberg, L. O. B. 1985. Effect of ammonia on glutamate, GABA and rubidium uptake by astrocytes. Ann. Neurol. 18:149.
Booher, J., and Sensenbrenner, M. 1972. Growth and cultivation of dissociated neurons and glial cells from embryonic chick, rat, and human brain in flask cultures. Neurobiology 2:97–105.
Ducis, I., Norenberg, L. O. B., and Norenberg, M. D. 1990. The benzodiazepine receptor in cultured astrocytes from genetically epilepsy-prone rats. Brain Res. 531:318–321.
Stein, W. D. 1986. Transport and diffusion across cell membranes. Academic Press, San Diego.
Drejer, J., Larsson, O. M., and Schousboe, A. 1982. Characterization ofl-glutamate uptake into and release from astrocytes and neurons cultured from different brain regions. Exp. Brain Res. 47: 259–269.
Bender, A. S., Woodbury, D. M., and White, H. S. 1989. β-DL-Methylene aspartate, an inhibitor of aspartate aminotransferase, potently inhibitsl-glutamate uptake into astrocytes. Neurochem. Res. 14:641–646.
Goldschmidt, R. C., and Kimelberg, H. K. 1989. Protein analysis of mammalian cells in monolayer culture using the bicinchoninic assay. Anal. Biochem. 177:41–45.
Zivin, J. A., and Waud, D. R. 1982. How to analyze binding, enzyme and uptake data: the simplest case, a single phase. Life Sci. 30:1407–1422.
Gregorios, J. B., Mozes, L. W., Norenberg, L. O. B., and Norenberg, M. D. 1985. Morphologic effects of ammonia on primary astrocyte cultures. I. Light microscopic studies. J. Neuropathol. Exp. Neurol. 44:397–403.
Gregorios, J. B., Mozes, L. W., and Norenberg, M. D. 1985. Morphologic effects of ammonia on primary astrocyte cultures. II. Electron microscopic studies. J. Neuropathol. Exp. Neurol. 44: 404–414.
Norenberg, M. D., Baker, L., Norenberg, L. O. B., Blicharska, J., Bruce-Gregorios, J. H., and Neary, J. T. 1991. Ammonia-induced astrocyte swelling in primary culture. Neurochem. Res. 19:833–836.
Butterworth, R. F., Lavoie, J., Giguere, J. F., Pomier Layrargues, G., and Bergeron, M. 1987. Cerebral GABA-ergic and glutamatergic function in hepatic encephalopathy. Neurochem. Pathol. 6: 131–144.
Fan, P., and Szerb, J. C. 1993. Effects of ammonia ions on synaptic transmission and on responses to quisqualate and N-methyl-D-aspartate in hippocampal CA1 pyramidal neurons in vitro. Brain Res. 632:225–231.
Mena, E. E., and Cotman, C. W. 1985. Pathologic concentrations of ammonium ions blocks L-glutamate uptake. Exp. Neurol. 89: 259–263.
Rao, V. L. R., and Murthy, C. R. K. 1991. Hyperammonemic alterations in the uptake and release of glutamate and aspartate by rat cerebellar preparations. Neurosci. Lett. 130:49–52.
Huang, R., Kala, G., Murthy, Ch. R. K., and Hertz, L. 1994. Effects of chronic exposure to ammonia on glutamate and glutamine interconversion and compartmentation in homogeneous primary cultures of mouse astrocytes. Neurochem. Res. 19:257–265.
Waniewski, R. A. 1992. Physiological levels of ammonia regulate glutamine synthesis from extracellular glutamate in astrocyte cultures. J. Neurochem. 58:167–174.
Hertz, L., Murthy, Ch. R. K., Lai, J. C. K., Fitzpatrick, S. M., and Cooper, A. J. L. 1987. Some metabolic effects of ammonia on astrocytes and neurons in primary cultures. Neurochem. Pathol. 6: 97–129.
Dombro, R. S., Bender, A. S., Hutson, D. G., and Norenberg, M. D. 1995. Intracellular glutamine as a factor in ammonia-induced astrocyte swelling. Soc. Neurosci. Abstr. 21:1081.
Blei, A. T., Olafsson, S., Therrien, G., and Butterworth, R. F. 1994. Ammonia-induced brain edema and intracranial hypertension in rats after portacaval anastomosis. Hepatalogy 19:1437–1444.
Takahashi, H., Koehler, R. C., Brusilow, S. W., and Traystman, R. J. 1991. Inhibition of brain glutamine accumulation prevents cerebral edema in hyperammonemic rats. Am. J. Physiol. 261: H825-H829.
Jessy, J., Mans, A. M., DeJoseph, M. R., and Hawkins, R. A. 1990. Hyperammonaemia causes many of the changes found after portacaval shunting. Biochem. J. 272:311–317.
Albrecht, J., and Norenberg, M. D. 1990.l-Methionine-DL-sulfoximine induces massive efflux of glutamine from cortical astrocytes in primary culture. Eur. J. Pharmacol. 182:587–590.
Norenberg, M. D., and Bender, A. S. 1994. Astrocytic swelling in liver failure: role of glutamine and benzodiazepines. Acta Neurochir (Suppl.) 60:24–27.
Schmidt, W., Wolf, G., Grüngreiff, K., Meier, M., and Reum, T. 1990. Hepatic encephalopathy influences high-affinity uptake of transmitter glutamate and aspartate into the hippocampal formation. Metab. Brain Dis. 5:19–31.
Marcaida, G., Felipo, V., Hermenegildo, C., Minana, M.-D., and Grisolia, S. 1992. Acute ammonia toxicity is mediated by the NMDA type of glutamate receptors. FEBS Lett. 296:67–68.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bender, A.S., Norenberg, M.D. Effects of ammonia onl-glutamate uptake in cultured astrocytes. Neurochem Res 21, 567–573 (1996). https://doi.org/10.1007/BF02527755
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02527755