Abstract
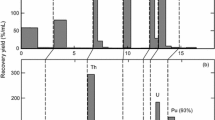
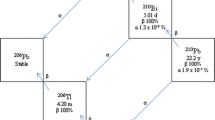
Using as eluent a sequence of 3M HCl, 12M HCl, and 8M HNO3, a mixture of210Pb,210Bi, and210Po may be clearly separated on a column of Dowex 1×2−100 anion exchange resin. A Cherenkov count in H2O and the variation in count rate with time confirm that the nuclides emerge in the order210Pb→210Bi→210Po. If 12M HCl is replaced by 1.5M H2SO4/2.3 M Na2SO4, a clean separation also results, but recovery of210Po becomes considerably more difficult. All three nuclides are readily detectable by liquid scintillation counting, with the efficiency for210Pb in the 60–70% range. The Cherenkov aqueous counting efficiency for210Bi is ∼14–15%.
Similar content being viewed by others
References
J. RUZICKA, J. STARY, Talanta, 8 (1961) 228.
W. D. FAIRMAN, J. SEDLET, Anal. Chem., 40 (1968) 2004.
H. BATEMAN, Cambridge Phil. Soc. Proc., 15 (1910) 423.
T. ISHIMORI, Bull. Chem. Soc. Japan, 28 (1955) 432.
E. D. BRANSOME, Jr. (Ed.), The Current Status of Liquid Scintillation Counting, Grune and Stratton, New York, 1970.
K. W. BAGNALL, The Chemistry of Selenium, Tellurium, and Polonium, Elsevier, Amsterdam, 1966.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pacer, R.A. The role of Cherenkov and liquid scintilation counting in evaluating the anion-exchange separation of210Pb−210Bi−210Po. J. Radioanal. Chem. 77, 19–28 (1983). https://doi.org/10.1007/BF02525349
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02525349