Abstract
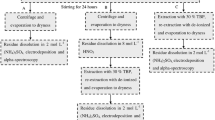
A simple method is presented for the determination of inorganic arsenic in water samples. The lower limit of determination is 0.01 μg·1−1. Penta-and trivalent arsenic ions as well as arsenic acid substituted with aromatic ligands are collected by coprecipitation with Fe(OH)3. After irradiation the arsenic is separated from the iron by adsorption to Al2O3. The overall efficiency is (97∓1)%. The procedure takes one day per ∼20 samples. It has been checked by standard addition. Data for sea- and surface-water are given. As the concentration of aromatic arsenic-compounds is very small in comparison to that of the inorganic species, it does not affect the result of the determination.
Similar content being viewed by others
References
R. S. BRAMAN, et al., Anal. Chem., 49 (1977) 621.
M. O. ANDREAL, Anal. Chem., 49 (1977) 820.
A. V. SHAIKH, D. E. TALLMAN, Anal. Chim. Acta, 98 (1978) 251.
P. SCHILLER, et al., Anal. Chim. Acta, 54 (1971) 364.
V. I. PLOTNIKOV, Zh. Analit. Khim., 19 (1964) 1183.
Y. S. SHEN, J. A. W. W. A. 65 (1973) 543.
M. ISHIBASHI, et al., Rec. of Ocean Works of Japan, 5 (1960) 66.
R. PIERUCCINI, Spectrochim. Acta, 4 (1950) 189.
G. D. CHRISTMAN, F. J. FELDMAN, Atomic Absorption Spectroscopy, Wiley Interscience, New York, 1970, p. 417.
F. GIRARDI, Report UUR 4287C.
L. A. CURRIE, Anal. Chem., 40 (1968) 586.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wals, G.D., Das, H.A. & Van Der Sloot, H.A. The determination of inorganic arsenic in water by thermal neutron activation analysis. J. Radioanal. Chem. 57, 215–222 (1980). https://doi.org/10.1007/BF02519881
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02519881