Abstract
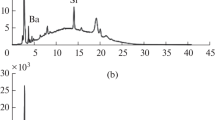
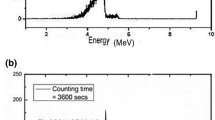
A method has been developed for the direct determination of uranium in carbonate solutions by X-ray fluorescence. Uranium, in the concentration range 0.2–50 ppm, is first absorbed as the carbonate complex on an anion exchange resin and then determined by X-ray fluorescence. A sensitivity corresponding to 20 ppb in 250 ml solution and a precision of ±6.5% was obtained. The sensitivity was 100-fold greater than that obtained by the direct determination in solution by X-ray fluorescence.
Similar content being viewed by others
References
S. AMIEL, Anal. Chem., 34 (1962) 13.
J. F. KLECKA, Univ. of California, Lawrence Radiation Laboratory Report UCRL-17144 Rev. Oct. 1966.
J. O. KARTTUNEN, H. R. HARMON, Spectrochim. Acta, 24B (1969) 301.
C. SHENBERG, S. AMIEL, Anal. Chem., 43 (1971) 1025.
R. J. N. BRITS, M. C. B. SMIT, Anal. Chem., 49 (1977) 67.
R. J. N. BRITS, H. A. DAS, Radiochem. Radioanal. Lett., 35 (1978) 6.
L. R. HATHAWAY, G. W. JAMES, Anal. Chem., 47III (1975) 2035.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Feldstein, H., Gilath, I. Determination of low uranium concentration in carbonate-bicarbonate solutions by X-ray fluorescence. J. Radioanal. Chem. 57, 47–52 (1980). https://doi.org/10.1007/BF02519863
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02519863