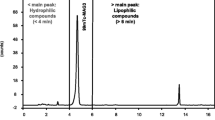
Abstract
Radio high pressure liquid chromatography (radio-HPLC) is the method of choice for quality control of radiopharmaceuticals labelled with short lived isotopes. Our preparations of “no carrier added”11C-labelled palmitic acid and L-methionine are both designed to end with a HPLC separation on either silica gel or C-18 reversed phase material. Since the crude reaction mixtures contain milligram amounts of inactive substrate materials, both separations must be carried out at preparative scale. Nevertheless they are performed in less than 10 min. The most critical factor for the separation of11C-palmitic acid from the main by-product pentadecane is the solvent composition: while the11C-L-methionine separation is especially sensitive to pH variations.
Similar content being viewed by others
References
D. R. CHRISTMAN, R. D. FINN, K. I. KARLSTROM, A. P. WOLF, Int. J. Appl. Radiat. Isotopes, 26, (1975) 435.
G.-J. MEYER, T. HARMS, H. HUNDESHAGEN, Proc. 4 th Int. Symp. Radiopharm. Chem., Aug. 23–27, 1982, Jülich, F. R. Germany, p. 92. See also J. Labelled Comp. Radiopharm., 19 (1982) 1362.
H.-J. MACHULLA, G. STÖCKLIN, CH. KUPFERNAGEL, CH. FREUNDLIEB, A. HÖCK, K. VYSKA, L. E. FEINENDEGEN, J. Nucl. Med., 19, (1978) 298.
M. J. WELCH, C. S. DENCE, M. R. KILBOURN, Proc. 4 th Int. Symp. Radiopharm. Chem., Aug. 23–27, 1982, Jülich, F. R. Germany, p. 112. See also J. Labelled Comp. Radiopharm., 19 (1982) 1382.
P.D. SCHUBIGER, M. ARGENTINI, Proc. 19th Int. Annual Meeting Soc. Nucl. Med., Europe, Sept. 8–11, 1981, Bern, Switzerland, p. 139.
G. BERGER, M. MAZIERE, R. KNIPPER, C. PRENANT, D. COMAR, Int. J. Appl. Radiat. Isotopes 30, (1979) 393.
G.-J. MEYER, A. OSTERHOLZ, H. HUNDESHAGEN, Proc. 4th Int. Symp. Radiopharm. Chem., Aug. 23–27, 1982, Jülich, F. R. Germany. See also J. Labelled Comp. Radiopharm., 19 (1982) 1286.
G.-J. MEYER, Radiochimica Acta, 30, (1982) 175.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Meyer, G.J., Osterholz, A. & Hundeshagen, H. Routine quality control of11C-labelled radiopharmaceuticals by high pressure liquid chromatography. J. Radioanal. Chem. 80, 229–235 (1983). https://doi.org/10.1007/BF02517667
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02517667