Abstract
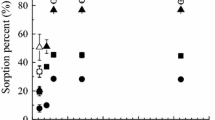
The sorption of144Ce(III) and of147Pm(III) on hydrated iron sesquioxide suspension and the sorption of144Ce(III) on hydrated iron sesquioxide in a laboratory column were studied. The dependence of the sorption on pH and time, the sorption isotherm, the temperature-dependence of sorption, and the effects of ionic strength on sorption and desorption were determined under static conditions. The maximum sorptions of144Ce(III) and147Pm(III) were reached at pH=7.4 (Kd=8.9·105) and pH=9.1 (Kd=6.2·105), respectively.
Similar content being viewed by others
References
C. B. Amphlett, Neorganicheskie ionity, Mir, Moscow, 1966, p. 113.
V. Veselý, V. Pekárek, Talanta, 19 (1972) 219.
M. J. Fuller, Chromatogr. Rev., 14 (1071) 45.
C. B. Amphlett, L. A. McDonald, M. J. Redman, J. Inorg. Nucl. Chem., 6 (1958) 236.
N. B. Micheev, L. M. Micheeva, Dokl. Akad. Nauk SSSR, 158 (1964) 440.
A. Kozawa, J. Electrochem. Soc., 106 (1959) 552.
P. J. Anderson, Trans. Farady Soc., 54 (1957) 54.
A. K. Covington, T. Cressey, B. G. Lever, H. R. Thirsk, Trans. Faraday Soc., 58 (1972) 1975.
F. Kepák, Coll. Czech. Chem. Commun., 31 (1966) 3051.
Z. Kolařík, Coll. Czech. Chem. Commun., 27 (1962) 938.
F. Kepák, J. Křivá, J. Inorg. Nucl. Chem., 33 (1971) 1741.
F. M. Nelsen, F. T. Eggertsen, Anal. Chem., 30 (1958) 1387.
F. Karas, in Ion Exchangers, their Properties and Application (Ed J. Šmid), SNTL Prague 1954, p. 583.
Yu. V. Jegorov, Radiokhimiya, 14 (1971) 183.
T. Moeller, H. E. Kremers, Chem. Rev., 37 (1945) 97.
A. Breeuwsma, J. Lyklema, Disc. Farady Soc. No. 52 Surface Chemistry of Oxides 1971, p. 276.
V. Kellő, A. Tkáč, Physical Chemistry, ALFA, Bratislava 1969, p. 566.
H. A. Laitinen, Chemical Analysis, New York-Toronto-London, 1960, p. 169.
R. Griessbach, Theory and Practice of Ion Exchange Inostr. Lit., Moscow, 1957, p. 195.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kepák, F., Nuderová, M. & Kaňka, J. Sorption of144Ce(III) and147Pm(III) on hydrated iron sesquioxide. J. Radioanal. Chem. 14, 325–333 (1973). https://doi.org/10.1007/BF02516596
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02516596