Abstract
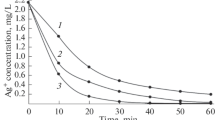
The surface properties of titanium hydroxide precipitates, formed under different conditions were studied in the presence of carrier-free radioactive89Sr. It was found that titanium with concentrations equivalent to 24 mg TiO2/litre, was enough to remove by adsorption more than 95% of the carrier-free strontium activity from an alkaline solution (pH≈13) in less than 6 hrs, regardless of whether the precipitate was freshly prepared or aged for a few days. If the precipitate was formed in situ, at the above pH value, the percentage removal of the strontium activity from the solution was even greater, and this was attributed to an internal adsorption process. At a lower pH value (∼2.5), however, the adsorption of strontium activity by titanium hydroxide was very low (<5%), whether the precipitate was preformed or formed in situ. The increase in the adsorption ability of titanium hydroxide precipitate on going from low pH (2. 5) to higher values was attributed to the inversion of the charge of titanium hydroxide sols from cationic particles at pH 2. 5 to negatively-charged particles at pH>3. 5, these latter particles adsorbing any positive strontium ions in solution.
Similar content being viewed by others
References
T. Komatsu, J. Water Pollut. Contr. Fed., 39 (1967) 123.
H. W. Levi, E. Schiewer, Radiochim. Acta, 14 (1970) 43.
H. W. Levi, Proc. Symp. on Treatment and Storage High-Level Radioactive Wastes, IAEA, Vienna, 1962, p. 587.
J. Lefevre, Energie Nucl., 8 (1966) 86.
N. K. Ghattas, Thesis, Ain Shams University, Cairo, Egypt, 1972.
G. Goldstein, Nucleonics, 23 (1965) 67.
O. Hahn, Applied Radiochemistry, Cornell Univ. Press, Ithaca, N. Y., 1936.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
El-Atrash, A.M., Azzam, A.M. & Ghattas, N.K. Study of the surface properties of titanium hydroxide using a radioactive tracer technique. J. Radioanal. Chem. 23, 17–22 (1974). https://doi.org/10.1007/BF02514340
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02514340