Abstract
Morphological and structural studies carried out inXanthoria parietina reveal some fungal mechanisms to regulate both growth and development of the phycobiont as well as the number of photobiont cells present in the holobiont. This regulation is performed by phenolic acids and glycosyl-enzymes.
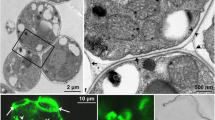
An ultrastructural analysis using the polysaccharide detection technique PATAg shows that plasmolysis of cells occur when freshly isolated phycobionts are incubated with two arginase lectins (ABP, algal binding protein and SA, secreted arginase), with development of large cytoplasmic vesicles filled with amorphous polysaccharides that are exocyted to the periplasmic space. Finally, membranes of organelles and plasma membrane are altered and the cell wall is broken.
The results presented here provide evidence of a possible fungus-to-algal action as deduced from the hemiparasitic symbiosis theory.
Similar content being viewed by others
Abbreviations
- SA:
-
secreted arginase
- ABP:
-
algal binding protein
- PATAg:
-
periodique acide-thiocarbohydrazide-argent
- PBS:
-
phosphate saline buffer
References
Ahmadjian, V. 1982. Algal/fungal symbioses.In F.J. Round and D.J. Chapman, eds., Progress in Phycological Research, Elsevier Biomedical Press, Amsterdam, pp. 179–233.
Ascaso, C. andGalván, J. 1976. The ultrastructure of the symbionts ofRhizocarpon geographicum, Parmelia conspersa andUmbilicaria pustulata growing under dryness conditions. Protoplasma87: 409–418.
Avalos, A. andVicente, C. 1987. The occurrence of lichen phenolics in the photobiont cells ofEvemia prunastri. Plant Cell Reports6: 74–76.
Boissiere, M.C. 1982. Cytochemical ultrastructure ofPeltigera canina: some features related to its symbiosis. Lichenologist14: 1–27.
Honegger, R. 1986. Ultrastructural studies in lichens. II. Mycobiont and photobiont cell wall surface layers and adhering crystalline lichen products in fourParmeliaceae. New Phytol.103: 797–808.
Honegger, R. 1987. Questions about pattern formation in the algal layer of lichens with stratified (heteromerous) thalli. Bibl. Lichenol.25: 59–71.
Honegger, R. 1991. Functional aspects of the lichen symbiosis. Annu. Rev. Plant Physiol. Plant Mol. Biol.42: 553–578.
Honegger, R. andBrunner, R. 1981. Sporopollenin in the cell walls ofCoccomyxa andMyrmecia phycobionts of various lichens: an ultrastructural and chemical investigation. Can. J. Bot.59: 2713–2734.
Hill, D.J. 1989. The control of the cell cycle in microbial symbionts. New Phytol.112: 175–184.
Hill, D.J. 1992. The co-ordination of development of symbionts in mutualistic symbiosis with reference to the cell cycle of the photobiont in lichens. Symbiosis14: 325–333.
Hill, D.J. 1994. The cell cycle of the photobiont of the lichenParmelia sulcata (Lecanorales, Ascomycotina) during development of the thallus lobes. Crypt. Bot.4: 270–273.
Mac Manus, J.F.A. 1948. Histological and histochemical uses of periodic acid. Stain Technol.23: 99–108.
Molina, M.C., Muñiz, E. andVicente, C. 1993a. Enzymatic activities of algal-binding protein and its algal cell wall receptor in the lichenXanthoria parietina. An approach to the parasitic basic of mutualism. Plant Physiol. Biochem.32: 131–142.
Molina, M.C., Stocker-Wörgötter, E., Zorer, R., Türk, R. andVicente, C. 1997. Comparison between recent-isolated and cultured natural populations of photobionts fromXanthoria parietina.In H.E.A. Schenck, R.G. Hermann, K.W. Jeon, N.E. Muller and W. Schwemmler, eds., Eukaryotism and Symbiosis, Springer-Verlag, Berlin, Heidelberg, pp. 484–490.
Molina, M.C. andVicente, C. 1993. Loss of photoergonic condition ofXanthoria parietina photobiont effected by an algal-binding protein isolated from the same lichen species.In S. Sato, M. Ishida and H. Ishikawa, eds., Endocytobiology V, Tübingen University Press, Tübingen, pp. 69–74.
Molina, M.C. andVicente, C. 1995. Correlationship between enzymatic activity of lectins, putrescine content and chloroplast damage inXanthoria parietina phycobionts. Cell Adh. Com.3: 1–12.
Molina, M.C. andVicente, C. 1996. Ultrastructural deterioration ofXanthoria parietina (L.) Th. Fr phycobiont induced by aXanthoria lectin. Phyton (Horn, Austria)36: 197–208.
Molina, M.C., Vicente, C. andMuñiz, E. 1993b. Cytochemical detection of urease activity in the cell walls of lichenXanthoria parietina and the effect of endogenous phenolics on urease activity. Acta Hort.381: 235–238.
Molina, M.C., Vicente, C., Pedrosa, M.M. andMuñiz, E. 1996. Binding of a labelled lectin from the lichenXanthoria parietina to its own phycobiont and analysis of this enzymatic activity. Phyton (Horn, Austria)36: 145–158.
Pedrosa, M.M. andLegaz, M.E. 1991. Binding studies of evernic acid to purified constitutive arginase ofEvernia prunastri by SE-HPLC. Symbiosis11: 345–357.
Reynolds, S. 1963. The use of lead citrate at high pH as and electron-opaque stain in electron microscopy. J. Cell. Biol.17: 200–211.
Thiéry, J.P. andRambourg, A. 1974. Cytochimie des polysaccharides. J. Microscopie21: 225–232.
Vian, B. andRoland, J.C. 1972. Différenciation des cytomembranes et renouvellement du plasmalemme dans les phénomènes de sécrétions végétales. J. Microscopie13: 119–136.
Vian, B. andRoland, J.C. 1991. Affinodetection of the sites of formation and of the further atribution of polygalacturonans and native cellulose in growing plant cells. Cell Biol.71: 43–55.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Molina, M.d.C., Bajon, C., Sauvanet, A. et al. Detection of polysaccharides and ultrastructural modification of the photobiont cell wall produced by two arginase isolectins fromXanthoria parietina . J. Plant Res. 111, 191–197 (1998). https://doi.org/10.1007/BF02512169
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02512169