Abstract
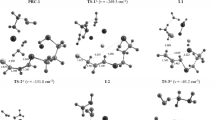
The sigmatropic and dissociative-recombination mechanisms of the chlorotropic isomerization of benzodioxachlorophosphazopentachloroethane and its 1:2 solvate with chloroform have been studied by the MNDO-PM3 method in the supermolecular approximation. It is observed that the thermodynamically more stable chlorotropic isomer corresponds to the phosphorane structure with axial-equatorial disposition of two chlorine atoms and the two oxygen atoms in the dioxaphosphole ring, in complete agreement with literature data on teh35Cl NQR spectrum. It is shown that the most likely pathway for phosphorus-carbon chlorotropic rearrangement for both phosphazopentachloroethane and its solvate with two molecules of chloroform is the sigmatropic route.
Similar content being viewed by others
References
E. A. Romanenko,Teor. Éksp. Khim.,35, No. 3, 155–161 (1999).
E. A. Romanenko,Teor. Éksp. Khim.,35, No. 4, 215–221 (1999).
V. I. Boiko, L. I. Samarii, N. V. Mel'nichenko, et al.,Zh. Org. Khim.,66, No. 10, 1715–1719 (1996).
J. J. P. Stewart,QCPE Program MOPAC 6.0 (1990).
E. M. Holt and S. L. Holt,J. Chem. Soc., Dalton Trans., 1990–1993 (1974).
T. S. Cameron and C. K. Prout,J. Chem. Soc. (C), 2281–2284 (1969).
E. A. Romanenko,Teor. Éksp. Khim.,25, No. 2, 237–240 (1989).
P. G. Perkins and J. J. P. Stewart,J. Chem. Soc. Faraday Trans. II,78, No. 2, 285–296 (1982).
Additional information
Institute of Bioorganic and Oil Chemistry, National Academy of Sciences of Ukraine, 1 Murmanskaya ul., Kiev 02094, Ukraine. Translated from Teoreticheskaya i Éksperimental'naya Khimiya, Vol. 36, No. 3, pp. 177–181, May–June, 2000.
Rights and permissions
About this article
Cite this article
Romanenko, E.A. Sigmatropic mechanism of the chlorotropic isomerization of phosphazopentachloroethane. Theor Exp Chem 36, 163–167 (2000). https://doi.org/10.1007/BF02511431
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02511431