Abstract
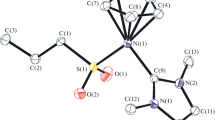
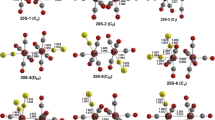
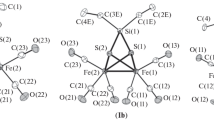
The reaction of the complex Re2(CO)4(NO)2Cl4 (1) with NaSCMe3 (2) (in THF or MeCN, 65–80°C, 24 h) was studied at different ratios of the reagents (from 1∶2 to 1∶6). At the reagent ratio of 1∶2, the binuclear complex Re2(CO)4(NO)2Cl2(μ-SCMe3)2 (3) was obtained as a mixture ofsyn andanti isomers (3a and3b, respectively) containing Re2S2 fragments with different structures (the butterfly-like structure in3a and the planar fragment in3b). When the initials were taken in ratios from 1∶4 to 1∶6, two compounds were isolated: the binuclear complex Re2(CO)4(NO)2(μ-SCMe3)2(μ-S)4 (cocrystallized as a mixture ofsyn andanti isomers,4a and4b, respectively) and the triangular cluster Re3(CO)3(NO)3(μ-SCMe3((μ3-S)(μ3-Cl) (5). Apparently, complex4 is formed in the course of isolation as a result of elimination of SR2 from the unstable tetrathiolate dimer Re2(CO)4(NO)2(SCMe3)2(μ-SCMe3)2 (6). Cluster5 is the product of the reaction between compounds3 and4. Products of interaction of compound6 with silica gel upon column chromatography of the reaction mixture were studied. Four complexes containing hydroxy and oxo bridging groups, (CO)2(NO)Re(μ-SCMe3)2(μ-OH)Re(SCMe3)(CO)(NO) (7), (CO)3(NO)3RE3(μ-SCMe3)3(μ3-SCME3)(μ3-O) (8), [(CO)2(NO)2Re2(SCMe3)2(μ-SCMe3)2(μ-OH)][Na(THF)(Et2O)] (9), and [(CO)2(NO)2Re2(SCMe3)2(μ-SCMe3)2(μ-OH)]2−[Na(H2O)6][H5O2] (10), were isolated. The structures of complexes3, 4, 5, 7, 8, 9, and10 were established by X-ray diffraction study.
Similar content being viewed by others
References
P. J. Blower and J. R. Dilworth,Coord. Chem. Rev., 1987,76, 121.
D. W. Rakowski,Chem. Rev., 1989,89, 1.
Metal Clusters in Proteins, ACS Symposium Ser., Ed. L. Qui, Jr., Am. Chem. Soc., New York, 1988, 372.
A. A. Pasynskii, I. L. Eremenko, S. B. Katser, A. A. Aliev, and M. A. Porai-Koshits,Metalloorg. Khim., 1990,2, 454 [Organomet. Chem. USSR, 1990,2 (Engl. Transl.)].
I. L. Eremenko, A. A. Pasynskii, S. E. Nefedov, A. S. Katugin, B. I. Kolobkov, A. D. Shaposhnikova, R. A. Stadnichenko, A. I. Yanovsky, and Yu. T. Struchkov,Zh. Neorg. Khim., 1992,37, 574 [Russ. J. Inorg. Chem., 1992,37 (Engl. Transl.)].
I. L. Eremenko, H. Berke, B. I. Kolobkov, and V. M. Novotortsev,Organometallics 1995,13, 244.
I. L. Eremenko, S. E. Nefedov, H. Berke, B. I. Kolobkov, and V. M. Novotortsev,Organometallics, 1995,14, 1132.
I. L. Eremenko, V. M. Novotortsev, I. A. Petrunenko, and H. Berke,Izv. Akad. Nauk, Ser. Khim., 1995, 2337 [Russ. Chem. Bull., 1995,44, 2233 (Engl. Transl.)].
F. Zingales, A. Trovati, F. Cariati, and P. Uguagliati,Inorg. Chem., 1971,10, 507.
H. Matsuzaka, Y. Hirayama, M. Nishio, Y. Mizobe, and M. Hidai,Organometallics, 1993,12, 36.
H. -U. Hund, Ph. D. Thesis, Zurich Univ., Zurich, 1992.
H. -U. Hund, U. Ruppli, and H. Berke,Helv. Chem. Acta, 1993,76, 963.
D. A. Veghini, Ph. D. Thesis, Zurich Univ., Zurich, 1995.
D. Veghini, S. E. Nefedov, H. Scmalle, and H. Berke,J. Organomet. Chem., 1996,526, 117.
I. L. Eremenko, V. I. Bakhmutov, F. Ott, and H. Berke,Zh. Neorg. Khim., 1993,38, 1653 [Russ. J. Inorg. Chem., 1993,38 (Engl. Transl.)].
A. A. Pasynskii, I. L. Eremenko, Yu. V. Rakitin, V. M. Novotortsev, V. T. Kalinnikov, G. G. Aleksandrov, and Yu. T. Struchkov,J. Organomet. Chem., 1979,165, 57.
E. A. Vastutinskaya, I. L. Eremenko, A. A. Pasynskii, S. E. Nefedov, A. I. Yanovsky, and Yu. T. Struchkov,Zh. Neorg. Khim., 1991,36, 1707 [Russ. J. Inorg. Chem., 1991,36 (Engl. Transl.)].
N. Walker and D. Sruart,Acta Crystallogr., 1983,A39, 158.
G. M. Sheldrick, inCrystallographic Computering 3. Data Collection, Structure Determination, Proteins, and Databases, Clarendon Press, New York, 1985, 175.
Author information
Authors and Affiliations
Additional information
Translated fromIzvestiya Akademii Nauk. Seriya Khimicheskaya, No. 5, pp. 1030–1044, May, 1998.
Rights and permissions
About this article
Cite this article
Nefedov, S.E., Sidorov, A.A., Berke, H. et al. Polynuclear nitrosyl carbonyl ReI complexes with thiolate and sulfide bridges. Russ Chem Bull 47, 1001–1015 (1998). https://doi.org/10.1007/BF02498175
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02498175