Abstract
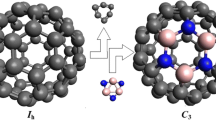
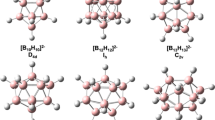
Molecular and electronic structure of heterofullerene BNC58 (C s) and B2N2C56 (C 2h) monomers, B2N2C116 and B4N4C112 dimers, and B6N6C168 trimer (the last three molecules withC 2h symmetry) was simulated by the MNDO method. Clusters BNC58 and B2N2C56 are formed by replacement of carbon atoms participating in one or two of the most distant oppositely lying (6,6)-type C−C bonds in fullerene C60 by B and N atoms. In one of the two studied isomers of the B2N2C116 dimer, the monomers are linked by the four-membered carbon cycle, while the heteroatoms form the most distant oppositely lying bonds of the dimer. In the other isomer of the B2N2C116 dimer, as well as in the B4N4C112 dimer and B6N6C168 trimer, the monomers are linked by four-membered B2N2 cycles with alternation of the atoms. For all the systems studied, the optimum geometric parameters, heats of formation, ionization potentials, and atomic charges were calculated. Dimerization energies of heterofullerenes BNC58 and B2N2C56 lie in the range from 33 to 49 kcal mol−1. It was found that the B2N2C116 dimer, in which the monomers are linked by the four-mernbered carbon cycle, is the most stable system. In the case of B2N2C56 trimerization, the energy gain (compared to the triple monomer energy) is about twice as large as the dimerization energy. Molecular structure of the quasi-linear [B2N2C56) n macromolecule was simulated, and extended Hückel calculations of its energy band structure by the crystal orbital method were performed. It was found that the electron energy spectrum is of semiconducting type (the band gap is equal to 1.27 eV.
Similar content being viewed by others
References
D. A. Bochvar and E. G. Gal'pern,Dokl. Akad. Nauk SSSR, 1973,209, 610 [Dokl. Chem., 1973 (Engl. Transl.)].
D. A. Bochvar and E. G. Gal'pern,Izv. Akad. Nauk SSSR, Ser. Khim., 1974, 2367 [Bull. Acad. Sci. USSR, Div. Chem. Sci., 1974,23 (Engl. Transl.)].
T. Guo, C. Jin, and R. E. Smalley,J. Phys. Chem., 1991,96, 4948.
N. Kurita, K. Kobayashi, H. Kumamora, and K. Tago,Phys. Rev. B, Condens. Matter, 1993,48, 4850.
T. Pradeep, V. Vijayakrishnan, A. K. Santra, and C. N. R. Rao,J. Phys. Chem., 1991,95, 10564.
J. C. Hummelen, B. Knight, J. Pavlovich, R. Gonzàlez, and F. Wudl,Science, 1995,269, 1554.
K. Prassides, M.-K. Keshavarz, J. C. Hummelen, W. Anderoni, P. Giannozzi, E. Beer, C. Bellavia, L. Cristofolini, R. Gonzàlez, A. Lappas, Y. Murata, M. Malecki, V. Strdanov, and F. Wudl,Science, 1996,271, 1833.
J. Piechota, P. Byszewski, R. Jablonski, and K. Antonova,Fullerene Sci. Technol., 1996,4, 491.
J. R. Bowser, D. A. Jelski, and T. F. George,Inorg. Chem., 1992,31, 156.
X. Xia, D. Jelski, J. R. Bowser, and T. F. George,J. Am. Chem. Soc., 1992,114, 6493.
P. W. Fowler, T. Heine, D. Mitchell, R. Schmidt, and G. Seifert,J. Chem. Soc., Faraday Trans., 1996,92, 2197.
K. Kobayashi and N. Kurita,Phys. Rev. Lett., 1993,70, 3542.
I. V. Stankevich, A. L. Chistyakov, and E. G. Gal'pern,Izv. Akad. Nauk, Ser. Khim., 1993, 1712 [Russ. Chem. Bull., 1993,42, 1364 (Engl. Transl.)].
I. V. Stankevich, A. L. Chistyakov, E. G. Gal'pern, and N. P. Gambaryan,Zh. Strukt. Khim., 1995,36, 976 [Russ. J. Struct. Chem., 1995,36 (Engl. Transl.)].
I. Silaghi-Dumitrescu, F. Lara-Ochoa, P. Bishof, and I. Haiduc,J. Mol. Struct. (Theochem.), 1996,367, 47.
O. Stephan, P. M. Ajayan, C. Colliex, Ph. Redlich, J. M. Lambert, P. Berkier, and P. Lefin,Science, 1994,226, 1683.
A. Loiseau, F. Willaime, N. Demoncy, G. Hug, and H. Pascard,Phys. Rev. Lett., 1996,76, 4737.
H. Y. Zhu, D. J. Klein, W. A. Seitz, and N. H. March,Inorg. Chem., 1995,34, 1377.
E. G. Gal'pern, I. V. Stankevich, L. A. Chernozatonskii, and V. V. Pinyaskin,J. Phys. Chem. B, 1997,101, 705.
Y. Miyamoto, A. Rubio, M. L. Cohen, and S. G. Louie,Phys. Rev. B, Condens. Matter, 1994,50, 4976.
W. Andreoni, A. Curioni, K. Holczer, K. Prassides, M.-K. Keshavarz, J. C. Hummelen, and F. Wudl,J. Am. Chem. Soc., 1996,118, 11335.
M. J. S. Dewar and W. Thiel,J. Am. Chem. Soc., 1977,99, 4899.
M. W. Schmidt, K. K. Baldridge, J. A. Boatz, S. T. Elbert, M. S. Gordon, J. H. Jensen, S. Koseki, N. Matsunaga, K. A. Nguyen, S. J. Su, T. L. Windus, M. Dupuis, and J. A. Montgomery,J. Comput. Chem., 1993,14, 1347.
R. Hoffmann,J. Chem. Phys., 1963,39, 1397.
D. A. Bochvar, E. G. Gal'pern, and I. V. Stankevich,Zh. Strukt. Khim., 1988,29, 26 [J. Struct. Chem. (USSR), 1988,29 (Engl. Transl.)].
E. G. Gal'pern, I. V. Stankevich, A. L. Chistyakov, and L. A. Chernozatonskii,Izv. Akad. Nauk, Ser. Khim., 1998, 7 [Russ. Chem. Bull., 1998,47, 1 (Engl. Transl.)].
S. Osawa, E. Osawa, and Y. Hirose,Fullerene Sci. Technol., 1995,3, 565.
K. Honda, E. Osawa, Z. Slanina, and T. Matsumoto,Fullerene Sci. Technol., 1996,4, 812.
E. G. Galperm, I. V. Stankevich, A. L. Chistyakov, and L. A. Chernozatonskii,Fulleren Sci. Technol., 1998,6, 499.
Author information
Authors and Affiliations
Additional information
Translated fromIzvestiya Akademii Nauk. Seriya Khimicheskaya, No. 3, pp. 431–435, March, 1999.
Rights and permissions
About this article
Cite this article
Gal'pern, E.G., Stankevich, I.V., Chistyakov, A.L. et al. Molecular and electronic structure of several heterofullerene BNC58 and B2N2C56 oligomers and [B2N2C56] n macromolecule. Russ Chem Bull 48, 428–432 (1999). https://doi.org/10.1007/BF02496155
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02496155