Abstract
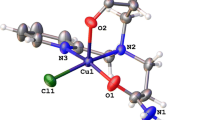
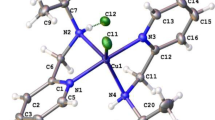
Copper(II) chelates of composition CuL2 were synthesized based on 4-aminomethylene derivatives of 5-thiopyrazoles (LH). The complexes were studied by UV, IR, ESR, and EXAFS spectroscopy, magnetochemistry, and X-ray diffraction analysis. The coordination polyhedra in the complexes are pseudotetrahedra or octahedra of the types CuN2S2 or CuN4S2, respectively, which are distorted due to the Jahn—Teller effect. The UV and ESR spectra of copper chelates with a six-coordinate metallocycle formed by the N and S atoms of the azomethine ligand and the nitrogen atom of the quinoline substituent (R) of the C=N−R fragment are most similar to the spectra observed for metals involved in the active centers of natural metalloenzymes (“blue” copper proteins).
Similar content being viewed by others
References
R. Malkin, inInorganic Biochemistry, Ed. G. Eichhorn, Elsevier, Amsterdam-Oxford-New York, 1978,2.
H. Toftlund, J. Becher, P. H. Olesen, and J. Pedersen,Israel J. Chem., 1985,25, 56.
E. T. Adman,Adv. Protein Chemistry, 1991,42, 144.
E. J. Solomon, M. J. Baldwin, and M. D. Lowery,Chem. Rev., 1992,92, 521.
S. Fox, J. A. Potenza, and S. Knapp, inBioinorganic Chemistry of Copper, Eds. K. D. Karlin and Z. Tucklar, Chapman and Hall, New York, 1993, 34.
S. J. Lippard and J. M. Berg,Principles of Bioinorganic Chemistry, California Univ. Sci. Books, 1994, 411.
W. Kaim and J. Rall,Angew. Chem., 1996,108, 47.
A. L. Nivorozhkin, A. I. Uraev, A. S. Burlov, and A. D. Garnovskii,Zh. Vsesovuz. Khim. Obshch. im. D. I. Mendeleeva, 1996,40, 162 [Mendeleev Chem. J., 1996,40, 255 (Engl. Transl.)].
S. Madal, G. Das, R. Singh, R. Shukla, and P. K. Bharadwaj,Coord. Chem. Rev., 1997,160, 191.
A. G. Sykes,Adv. Inorg. Chem., 1991,36, 377.
N. Kitajima, K. Fujisawa, M. Tanaka, and V. Moto-oka,J. Am. Chem. Soc., 1992,114, 9232.
D. Qui, L. T. Kilpatrick, N. Kitajima, and S. T. Spiro,J. Am. Chem. Soc., 1994,116, 2585.
N. Kitajima and W. R. Tolman,Progr. Inorg. Chem., 1995,42, 419.
A. D. Garnovskii, A. L. Nivorozhkin, and V. I. Minkin,Coord. Chem. Rev., 1993,126, 1.
A. E. Misdryukov, I. S. Vasil'chenko, V. S. Sergienko, A. L. Nivorozkin, S. G. Kochin, M. A. Poraj-Koshits, L. E. Nivorozhkin, and A. D. Garnovskii,Mendeleev Commun., 1992,30.
L. N. Kurkovskaya, N. N. Shapet'ko, I. Ya. Kvitko, Yu. N. Koshelev, and E. D. Samartseva,Zh. Org. Khim., 1974,10, 2210 [J. Org. Chem. USSR, 1974,10 (Engl. Transl.)].
W. Freyer and R. Redeglia,Monatsh. Chem., 1978,109, 1093.
I. Ya. Kvitko, L. V. Alam, M. N. Bobrovnikov, G. V. Avetikyan, and S. L. Panasyuk,Zh. Obshch. Khim., 1994,64, 657 [Russ. J. Gen. Chem., 1994,64 (Engl. Transl.)].
A. S. Antsyshkina, G. G. Sadikov, A. I. Uraev, and A. D. Garnovskii,Kristallografiya, 2000,45, No. 5 [Crystallogr. Repts., 2000,45, No. 5 (Engl. Transl.)].
V. I. Minkin, A. D. Garnovskii, J. Elguero, and A. R. Katritzkii, inThe Tautomerism of Heterocyclic Compounds. Acad. Press, San Diego, 2000,76, 153.
A. D. Garnovskii, V. P. Kurbatov, B. A. Porai-Koshits, O. A. Osipov, I. Ya. Kvitko, L. S. Minkina, E. M. Sof'ina, and A. F. Soloshko-Doroshenko,Zh. Obshch. Khim. 1970,40, 2338 [J. Gen. Chem. USSR, 1970,40 (Engl. Transl.)].
A. L. Nivorozhkin, M. S. Korobov, L. E. Konstantinovskii, L. E. Nivorozhkin, and V. I. Minkin,Zh. Obshch. Khim., 1985,55, 849 [J. Gen. Chem. USSR, 1985,55 (Engl. Transl.)].
A. I. Uraev, V. P. Kurbatov, and A. D. Garnovskii,Koord. Khim., 1997,23 [Russ. J. Coord. Chem., 1997,23 (Engl. Transl.)].
A. I. Uraev, A. L. Nivorozhkin, and A. S. Antsyshkina,Dokl. Akad. Nauk, 1997,356, 212 [Dokl. Chem., 1997 (Engl. Transl.)].
A. L. Nivorozhkin, A. I. Uraev, G. I. Bondarenko, A. S. Antsycshkina, V. P. Kurbatov, C. I. Turte, and N. D. Brashovianu,J. Chem. Soc., Chem. Commun., 1997, 1711.
V. P. Kurbatov, A. V. Khokhlov, A. D. Garnovskii, O. A. Osipov, and L. A. Khukhlachieva,Koord. Khim., 1979,5, 351 [Sov. J. Coord. Chem., 1979,5 (Engl. Transl.)].
D. I. Kochubei, Yu. A. Babanov, and K. I. Zamaraev,Rentgenospektral'nyi metod izucheniya amorfnykh tel. EXAFS-spektroskopiya [X-ray Spectral Method in Studies of Amorphous Substances, EXAFS Spectroscopy]. Nauka, Siberian Branch, Novosibirsk, 1988, 306 pp. (in Russian).
A. T. Shuvaev, B. Yu. Khel'mer, and T. A. Lyubeznova,Pribory i tekhnika eksperimenta [Instruments and Experimental Techniques], 1988,3, 234 (in Russian).
S. I. Zabenski, J. J. Rehr, A. Ancudinov, R. C. Albers, and M. J. Eller,Phys. Rev.,52, 2995.
G. M. Sheldrick,SHELXTL Plus. PC Version, A. System of Computer Programs for the Determination of Crystal Structure from X-Ray Diffraction Data. Rev. 502, Siemens Analitical X-ray Instruments Inc., Germany, 1994.
B. A. Porai-Koshits and I. Ya. Kvitko,Zh. Obshch. Khim., 1962,32, 4050 [J. Gen. Chem. USSR, 1962,32 (Engl. Transl.)].
Author information
Authors and Affiliations
Additional information
Published inIzvestiya Akademii Nauk. Seriya Khimicheskaya, No. 11, pp. 1891–1896, November, 2000.
Rights and permissions
About this article
Cite this article
Uraev, A.I., Nivorozhkin, A.L., Bondarenko, G.I. et al. Synthesis, structures, and spectral properties of biomimetic azomethine metal chelates with chromophores CuN2S2, CuN2O2, and CuN2Se2. Crystal structure of bis[4-(benzyl)aldimino-3-methyl-1-phenyl-5-pyrazolothiolato]copper(II). Russ Chem Bull 49, 1863–1868 (2000). https://doi.org/10.1007/BF02494925
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02494925