Abstract
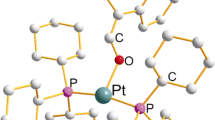
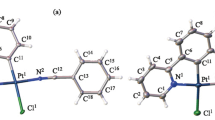
1:1 and 1:2 complexes of Ph2P(CH2)2P(S)Ph2 with PdCl2 were synthesized. Their structures were established by31P NMR and IR spectroscopy and X-ray diffraction analysis. In the crystals, the 1:1 complex has a chelate structure. In CH2Cl2, this complex partially dissociates at the Pd.S=P bond. According to the X-ray diffraction data, only the PIII atoms the cationic chelate complex [Pd(Ph2P(CH2)2P(S)Ph2)2]2+ 2NO3 −.
Similar content being viewed by others
References
S. Singh, N. K. Jha, P. Narula, and T. P. Singh,Acta Crystallogr., Sect. C., 1995,51, 593.
P. Groth,Acta Chem. Scand., 1970,24, 2785.
L. M. Engelhardt, J. M. Patrick, C. L. Raston, P. Twiss, and A. H. White,Aust. J. Chem., 1984,37, 2193.
A. Dohring, R. Goddard, I. Hopp, P. W. Jollg, N. Koket, and C. Kruger,Inorg. Chim. Acta, 1994,222, 179.
R. U. Kirss and R. Eisenberg,Inorg. Chem., 1989,25, 3372.
T. C. Blagborough, R. Davis, and P. Ivison,J. Organomet. Chem., 1994,467, 85.
T. E. Kron, A. N. Soboleva, M. I. Terekhova, N. A. Bondarenko, E. S. Petrov and E. N. Tsvetkov,Koord. Khim., 1992,18, 103 [Russ. J. Coord. Chem., 1992,18 (Engl. Transl.)].
J. A. Davies, A. A. Pinkerton, and M. Vilmer,Acta Crystallogr., Sect. C., 1992,48, 1027.
T. S. Lobana, M. K. Sandhu, M. J. Liddell, and E. R. T. Tiekink,J. Chem. Soc., Dalton Trans, 1990, 691.
M. J. Baker, M. F. Gioles, A. G. Orpen, M. J. Taylor, and R. J. Watts,J. Chem. Soc., Chem. Commun., 1995, 197.
E. I. Matrosov, Z. A. Starikova, A. I. Yanovsky, D. I. Lobanov, I. M. Aladzheva, O. V. Bykhovskaya, Y. T. Struchkov, T. A. Mastryukova, and M. I. Kabachnik,J. Organomet. Chem., 1997,535, 121.
A. Weigt and S. Bishoff,Main Group Chem. News, 1995,3, 11.
C. Abu-Gnim and I. Amer,J. Chem. Soc., Chem. Commun., 1994, 115.
R. W. Wegman, A. G. Abatjoglou, and A. M. Harrison,J. Chem. Soc., Chem. Commun. 1987, 1891.
S. Choopani, R. Davis, K. Smith, and J. Tebby,Abstract of XIIIth International Conference on Phosphorus Chemistry, ICPC, Jerusalem, Israel, 1995, 223.
J. R. Dilworth, J. R. Miller, N. Wheatley, M. J. Baker, and J. G. Sunley,J. Chem. Soc. Chem. Commun., 1995, 1579.
L. C. Satek, H. L. Ammon, and I. M. Stewart,Acta Crystallogr. Sect. B, 1975,31, 2691.
M. F. Richardson,Acta Crystallogr., Sect. C., 1985,41, 27.
Yu. I. Zefirov and P. M. Zorkii,Usp. Khim., 1995,64, 448. [Russ. Chem. Rev., 1995,64 (Engl. Transl.)].
Structure Correlation, Eds. H.-B. Burgi and J. D. Dunitz,2, VCH, Weinheim, New York, 1994, 767 pp.
I. D. Kolosova, A. N. Soboleva, T. E. Kron, E. S. Petrov, and V. K. Bel'skii,Koord. Khim., 1986,12, 393 [Sov. J. Coord. Chem., 1986,12 (Engl. Transl.)].
E. I. Matrosov, V. A. Gilyarov, V. Yu. Kovtun, and M. I. Kabachnik,Izv. Akad. Nauk SSSR, Ser. Khim., 1971, 1162 [Bull. Acad. Sci. USSR, Div. Chem. Sci., 1971,20 (Engl. Transl.)].
F. A. Miller and C. H. Wilkins,Anal. Chem., 1952,24, 1253.
I. M. Aladzheva, O. V. Bykhovskaya, D. I. Lobanov, P. V. Petrovskii, M. Yu. Antipin, T. A. Mastryukova, and M. I. Kabachnik,Zh. Obshch. Khim., 1998,68, 1421 [Russ. J. Gen. Chem., 1998,68 (Engl. Transl.)].
S. J. Higgings, R. Taylor, and B. L. Shaw,J. Organomet. Chem., 1987,325, 285.
I. M. Aladzheva, O. V. Bykhovskaya, D. I. Lobanov, P. V. Petrovskii, T. A. Mastryukova, and M. I. Kabachnik,Zh. Obshch. Khim., 1995,65, 1585 [Russ. J. Gen. Chem., 199565 (Engl. Transl.)].
G. M. Sheldrick,SHELXTL, Version 5.0. Software Reference Manual, Siemens Industrial Automation, Inc., Madison, 1994.
Author information
Authors and Affiliations
Additional information
Translated fromIzvestiya Akademii Nauk. Seriya Khimicheskaya, No. 6, pp. 1122–1127, June, 2000.
Rights and permissions
About this article
Cite this article
Matrosov, E.I., Starikova, Z.A., Labanov, D.I. et al. P,S-Ligands. Complexes ofP,P,P′,P′-tetraphenylethylenediphosphine monosulfide with PdCl2 . Russ Chem Bull 49, 1116–1121 (2000). https://doi.org/10.1007/BF02494907
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02494907