Abstract
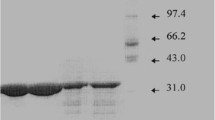
Phospholipase C (PLC; heat-labile hemolysin) was purified fromPseudomonas aeruginosa culture supernatants to near homogeneity by ion exchange chromatography. Some chelating reagents and divalent metal ions were examined for their effects on the purified PLC activity. EDTA, ECTA, ando-phenanthroline did not inhibit the enzyme activity top-nitrophenylphosphorylcholine (pNPPC) at concentrations below 10 mmol/L. Inclubation with Zn2+, Cu2+, Co2+ or Ni2+ caused a decrease in the enzyme activity. The activity of EDTA-treated PLC was most remarkably decreased in the presence of Zn2+, followed in descending order by Cu2+, Co2+ and Ni2+, while other metal ions (Mg2+, Ca2+, Ba2+, and Mn2+) did not significantly affect the enzyme activity using concentrations up to 10 mmol/L. Zn2+ competitively inhibited the enzyme activity against pNPPC, with a Ki value of 0.3 mmol/L. The hemolytic activity of PLC was also completely inhibited in the presence of 1 mmol/L Zn2+, and inactivation of the enzyme by Zn2+ was not reversed by concomtitant addition of other divalent metal ions. These results suggest that hemolytic PLC fromP. aeruginosa has a unique property in that there is no metal ion requirement for its activity.
Similar content being viewed by others
References
Liu PV. Toxins ofPseudomonas aeruginosa. In: Dogget RG (eds) Pseudomonas aeruginosa: clinical manifestations of infection and current therapy. New York: Academic Press, 1979:63–68.
Fujita K, Akino T, Yoshioka H. Characteristics of heartstable extracellular hemolysin fromPseudomonas aeruginosa. Infect Immun 1988;56:1385–1387.
Esselman M, Liu PV. Lectithinase production by Gramnegative bacteria. J Bacteriol 1961;81:939–945.
Coleman K, Dougan G, Arbuthnott JP. Oloning, and expression inEscherichia coli K12, of the chromosomal hemolysin (phospholipase C) determinat ofPseudomonas aeruginosa. J Bacteriol 1983;153:909–915.
Pritchard AE, Vasil ML. Neucleotide sequence and expresion of a phosphate-regulated gene encoding a secreted hemolysin ofPseudomonas aeruginosa. J Bacteriol 1986;167:291–298.
Ostroff RM, Vasil ML. Identification of a new phospholipase C activity by analysis of an insertional mutation in the hemolytic phospholipase C structural gene ofPseudomonas aeruginosa. J Bacteriol 1987;169:5497–4601.
Ostroff RM, Vasil AI, Vasil ML. Molecular comparison of a nonhemolytic and a hemolytic phospholipase C fromPseudomonas aeruginosa. J Bacteriol 1990;172:5915–5923.
Titball RW. Bacterial phospholipase C. Microbiol Rev 1993;57:347–366.
Berk RS, Brown D, Coutinho I, Meyers D. In vivo studies with two phospholipase C fractions fromPseudomonas aeruginosa. Infect Immun 1987;55:1728–1730.
Meyers DJ, Palmer KC, Bale LA, Kernacki K, Preston M, Brown T, et al. In vivo and in vitro toxicity of phospholipase C fromPseudomonas aeruginosa. Toxicology 1992;30:161–169.
Ostroff RM, Wretlind B, Vasil ML. Mutations in the hemolytic-phospholipase C operon result in decreased virulence ofPseudomonas aeruginosa PAO1 grown under phosphate-limiting conditions. Infect Immun 1989;57: 1369–1373.
Kurioka S, Matsuda M. Phospholipase C assay usingp-nitrophenylphosphorylcholine together with sorbitol and its application to studying the metal and detergent requirement of the enzyme. Anal Biochem 1976;75:281–289.
Krug EL, Kent C. Phospholipase C fromClostrudium perfringens: preparation and characterization of homogenous enzyme. Arch Biochem Biophys 1984;231:400–410.
Hough E, Hansen LK, Birkness B, Jynge K, Hansen S, Hordik A, et al. High resolution (1.5.A) crystal structure of phospholipase C fromBacillus cereus. Nature 1989;338: 357–360.
Geoffroy C, Raveneau J, Beretti JL, Lechroisey A, Vaquez-Boland JA, Alouf JE, et al. Purification and characterization of an extracellular 29-kilodalton phospholipase C fromListeria monocytogenes. Infect Immun 1991;59:2382–2388.
Titball RW, Rubidge T. The role of hisidine residues in the alpha toxin ofClostridium perfringens. FEMS Microbiol Lett 1990;68:261–266.
Berka RM, Vasil ML. Phospholipase C (heat-labile hemolysin) ofPseudomonas aeruginosa: purification and preliminary characterization. J Bacteriol 1982;152:239–245.
Chin JC, Watts JE. Blological propeties of phospholipase C purified from a fleecerot ofPseudomonas aeruginosa. J Gen Microbiol 1988;134:2567–2675.
Stinson MW, Hayden C. Secretion of phospholipase C byPseudomonas aeruginosa. Infect Immun 1979;25:558–564.
Berka RM, Gray GL, Vasil ML. Studies of phospholipase C (heart-labile hemolysin) inPseudomonas aeruginosa. Infect Immun 1981;34:1071–1074.
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976;72:248–254.
Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T. Nature 1970;227:680–685.
Dixon M, Webb EC. Enzymes. 3rd ed. London: Longman, 1979.
Sato H, Yamakawa Y, Ito A, Murata R. Effect of zinc and calcium ions on the production of alpha-toxin and proteases byClostridium perfringens. Infect Immun 1978;20:325–333.
Ikezawa H, Yamanegi M, Taguchi R, Miyashita T, Ohyabu T. Studies on phosphatidylinositol phosphodiesterase (phospholipase C type) ofBacillus cereus. II. Purification, properties and phosphatase-releasing activity. Biochim Biophys Acta 1976;450:154–164.
Taguchi R and Ikezawa H. Phospholipase C fromClostridium novyi type A. Biochim Biophys Acta 1975;409: 75–85.
Baine WB. A phospholipas C from the Dallas 1 E strain ofLegionella pneumophila serogroup 5: purification and characterization of conditions for optimal activity with an artificial substrate. J Gen Microbiol 1988;134:489–498.
Ikezawa H, Matsushita M, Tomita M, Taguchi R. Effects of metal ions on sphingomyelinase activity ofBacillus cereus. Arch Biochem Biophys 1986;249:588–595.
Author information
Authors and Affiliations
About this article
Cite this article
Tawara, S., Matsumoto, Y., Matsumoto, S. et al. Effect of metal lons on phospholipase C (Heat-Labile Hemolysin) fromPseudomonas aeruginosa . J Infect Chemother 3, 20–26 (1997). https://doi.org/10.1007/BF02489180
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02489180