Summary
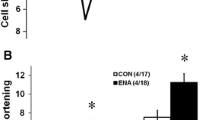
The purpose of this study was to examine the effects of the long-acting calcium channel antagonist pranidipine on ventricular remodeling, systolic and diastolic cardiac function, circulating humoral factors, and cardiac mRNA expression in myocardial infarcted rats. Myocardial infarction (MI) was produced by ligation of the coronary artery in Wistar rats. Three mg/kg per day of pranidipine was randomly administered to the infarcted rats. Hemodynamic measurements, Doppler echocardiographic examinations, analyses of the plasma levels of humoral factors, and myocardial mRNA expression were performed at 4 weeks after myocardial infarction. Left ventricular end-diastolic pressure (LVEDP) and central venous pressure (CVP) increased to 24.2 ± 1.2mmHg and 5.4 ± 0.6mmHg. Pranidipine reduced LVEDP and CVP to 13.6 ± 1.4mmHg (P < 0.01) and 2.5 ± 0.4mmHg (P < 0.01). The weight of the left and right ventricles in MI was significantly higher than in the sham-operated rats (sham, 2.02 ± 0.04 and 0.47 ± 0.02g/kg; MI, 2.18 ± 0.05 and 0.79 ± 0.04g/kg;P < 0.01). Left ventricular end-diastolic dimension (LVDd) in MI increased to 10.3 ± 0.3mm (P < 0.01) (sham, 6.4 ± 0.3mm). Pranidipine prevented an increase in the weight of the left and right ventricles (2.02 ± 0.04 and 0.6 ± 0.03g/kg,P < 0.01) and LVDd (7.9 ± 0.2mm,P < 0.01 to MI). Plasma renin activity (PRA), and plasma epinephrine, norepinephrine, and dopamine concentrations in MI were higher than those of the sham-operated rats. Pranidipine decreased the PRA and plasma cathecolamine levels of the myocardial infarcted rats to the level of the sham-operated rats. Moreover, the rats in MI showed systolic dysfunction, shown by decreased fractional shortening (sham, 31 ± 2% vs MI, 15 ± 1%;P < 0.01) and diastolic dysfunction shown by the E-wave deceleration rate (sham, 12.8 ± 1.1 m/s2; MI, 32.6 ± 2.1m/s2;P < 0.01). Pranidipine significantly prevented systolic and diastolic dysfunction. The increases in β-myosin heavy chain (MHC), α-skeletal actin, and atrial natriuretic polypeptide mRNAs in the noninfarcted left ventricle and right ventricle at 4 weeks after the myocardial infarction were significantly suppressed by the treatment with pranidipine. On the other hand, depressed α-MHC was restored to normal levels by pranidipine in both regions. In conclusion, pranidipine prevents the left ventricular remodeling process accompanied by systolic and diastolic dysfunction, and inhibits abnormal cardiac gene expression after myocardial infarction.
Similar content being viewed by others
References
Weisman HF, Bush DE, Mannisi JA, Bulkley BH (1985) Global cardiac remodeling after acute myocardial infarction: a study in the rat model. J Am Coll Cardiol 5:1355
McKay RG, Pfeffer MA, Pasternak RC, Markis JE, Come PC, Nakao S, Alderman JD, Ferguson JJ, Safian RD, Grossman W (1986) Left ventricular remodeling after myocardial infarction: a corollary to infarct expansion. Circulation 74:693
Pfeffer MA, Braunwald E (1990) Ventricular remodeling after myocardial infarction. Experimental observations and clinical implications. Circulation 81:1161
Yoshiyama M, Takeuchi K, Omura T, Kim S, Yamagishi H, Toda I, Teragaki M, Akioka K, Iwao H, Yoshikawa J (1999) Effects of candesartan and cilazapril on rats with myocardial infarction assessed by echocardiography. Hypertension 33:961
Pfeffer MA, Pfeffer JM, Steinberg C, Finn P (1985) Survival after an experimental myocardial infarction: beneficial effects of long-term therapy with captopril. Circulation 72:406
SAVE (1992) Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction. Results of the survival and ventricular enlargement trial. The SAVE Investigators. N Engl J Med 327:669
SOLVD (1991) Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. The SOLVD Investigators. N Engl J Med 325:293
CONSENSUS (1987) Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). The CONSENSUS Trial Study Group. N Engl J Med 316:1429
Packer M, O'Connor CM, Ghali JK, Pressler ML, Carson PE, Belkin RN, Miller AB, Neuberg GW, Frid D, Wertheimer JH, Cropp AB, DeMets DL (1996) Effect of amlodipine on morbidity and mortality in severe chronic heart failure. Prospective Randomized Amlodipine Survival Evaluation Study Group. N Engl J Med 335:1107
Shimada T, Yoshiyama M, Takeuchi K, Omura T, Takemoto Y, Kim S, Iwao H, Yoshikawa J (1998) Long acting calcium antagonist amlodipine prevents left ventricular remodeling after myocardial infarction in rats. Cardiovasc Res 37:618
Murdoch D, Heel RC (1991) Amlodipine. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic use in cardiovascular disease. Drugs 41:478
Little WC, Cheng CP (1994) Vascular versus myocardial effects of calcium antagonists. Drugs 4:41
Conti CR (1994) Use of calcium antagonists to treat heart failure. Clin Cardiol 17:101
Uehara Y, Kawabata Y, Hirawa N, Takada S, Numabe A, Matsuoka H, Ikeda T, Takabatake Y, Yagi S, Sugimoto T (1992) OPC-13340, a new dihydropyridine calcium channel blocker attenuates rapid vascular smooth muscle cell growth in spontaneously hypertensive rats. J Cardiovasc Pharmacol 20:408
Rosenthal J, Hittel N, Stumpe KO (1996) Pranidipine, a novel calcium antagonist, once daily, for the treatment of hypertension: a multicenter, double-blind, placebocontrolled dose-finding study. Cardiovasc Drugs Ther 10:59
Hirano T, Ohura M, Orito K, Fujiki H, Miyakoda G, Mori T (1997) Venodilator effects of pranidipine, a 1,4-dihydropyridine Ca2+ channel antagonist, in rats: comparison with nifedipine and amlodipine. Eur J Pharmacol 324:201
Nakayama N, Ikezono K, Mori T, Yamashita S, Nakayama S, Tanaka Y, Hosokawa T, Minami Y, Masutani K, Yamamura Y, Yabuuchi Y (1990) Antihypertensive activity of OPC-13340, a new potent and longacting dihydropyridine calcium antagonist, in rats. J Cardiovasc Pharmacol 15:836
Litwin SE, Katz SE, Morgan JP, Douglas PS (1994) Serial echocardiographic assessment of left ventricular geometry and function after large myocardial infarction in the rat. Circulation 89:345
Sahn DJ, DeMaria A, Kisslo J, Weyman A (1978) Recommendations regarding quantitation in M-mode echocardiography: results of a survey of echocardiographic measurements. Circulation 58:1072
Gay RG (1990) Early and late effects of captopril treatment after large myocardial infarction in rats. J Am Coll Cardiol 16:967
Kim S, Ohta K, Hamaguchi A, Yukimura T, Miura K, Iwao H (1995) Angiotensin II induces cardiac phenotypic modulation and remodeling in vivo in rats. Hypertension 25:1252
Genovese C, Rowe D, Kream B (1984) Construction of DNA sequences complementary to rat alpha 1 and alpha 2 collagen mRNA and their use in studying the regulation of type I collagen synthesis by 1,25-dihydroxyvitamin D. Biochemistry 23:6210
Liau G, Yamada Y, de Crambugghe B (1985) Coordinate regulation of the levels of type III and type I collagen mRNA in most but not all mouse fibroblasts. J Biol Chem 260:531
Kim S, Ohta K, Hamaguchi A, Omura T, Yukimura T, Miura K, Inada Y, Wada T, Ishimura Y, Chatani F, Iwao H (1994) Role of angiotensin II in renal injury of deoxycorticosterone acetate-salt hypertensive rats. Hypertension 24:195
MDPIT (1988) The effect of diltiazem on mortality and reinfarction after myocardial infarction. The Multicenter Diltiazem Postinfarction Trial Research Group. N Engl J Med 319:385
Gibson RS (1989) Current status of calcium channel-blocking drugs after Q wave and non-Q wave myocardial infarction. Circulation 80:107
Goldstein RE, Boccuzzi SJ, Cruess D, Nattel S (1991) Diltiazem increases late-onset congestive heart failure in postinfarction patients with early reduction in ejection fraction. The Adverse Experience Committee; and the Multicenter Diltiazem Postinfarction Research Group. Circulation 83:52
Iriarte M, Murga N, Sagastagoitia D, Molinero E, Morillas M, Salcedo A, Estella P, Etxebeste J (1993) Congestive heart failure from left ventricular diastolic dysfunction in systemic hypertension. Am J Cardiol 71:308
Gaasch WH (1994) Diagnosis and treatment of heart failure based on left ventricular systolic or diastolic dysfunction. JAMA 271:1276
Giannuzzi P, Imparato A, Temporelli PL, de-Vito F, Silva PL, Scapellato F, Giordano A (1994) Doppler-derived mitral deceleration time of early filling as a strong predictor of pulmonary capillary wedge pressure in postinfarction patients with left ventricular systolic dysfunction. J Am Coll Cardiol 23:1630
Xie GY, Berk MR, Smith MD, Gurley JC, DeMaria AN (1994) Prognostic value of Doppler transmitral flow patterns in patients with congestive heart failure. J Am Coll Cardiol 24:132
Hanatani A, Yoshiyama M, Kim S, Omura T, Toda I, Akioka K, Teragaki M, Takeuchi K, Iwao H, Takeda T (1995) Inhibition by angiotensin II type 1 receptor antagonist of cardiac phenotypic modulation after myocardial infarction. J Mol Cell Cardiol 27:1905
Chassagne C, Wisnewsky C, Schwartz K (1993) Antithetical accumulation of myosin heavy chain but not alphaactin mRNA isoforms during early stages of pressure-overload-induced rat cardiac hypertrophy. Circ Res 72:857
Schiaffino S, Samuel JL, Sassoon D, Lompre AM, Garner I, Marotte F, Buckingham M, Rappaport L, Schwartz K (1989) Nonsynchronous accumulation of alpha-skeletal actin and beta-myosin heavy chain mRNAs during early stages of pressure-overload-induced cardiac hypertrophy demonstrated by in situ hybridization. Circ Res 64:937
Swynghedauw B (1986) Developmental and functional adaptation of contractile proteins in cardiac and skeletal muscles. Physiol Rev 66:710
Hewett TE, Grupp IL, Grupp G, Robbins J (1994) Alphaskeletal actin is associated with increased contractility in the mouse heart. Circ Res 74:740
Izumo S, Nadal GB, Mahdavi V (1988) Protooncogene induction and reprogramming of cardiac gene expression produced by pressure overload. Proc Natl Acad Sci USA 85:339
Weber KT, Janicki JS, Shroff SG, Pick R, Chen RM, Bashey RI (1988) Collagen remodeling of the pressure-overloaded, hypertrophied nonhuman primate myocardium. Circ Res 62:757
Volders PG, Willems IE, Cleutjens JP, Arends JW, Havenith MG, Daemen MJ (1993) Interstitial collagen is increased in the non-infarcted human myocardium after myocardial infarction. J Mol Cell Cardiol 25:1317
Weber KT, Sun Y, Tyagi SC, Cleutjens JP (1994) Collagen network of the myocardium: function, structural remodeling and regulatory mechanisms. J Mol Cell Cardiol 26:279
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Takeuchi, K., Omura, T., Yoshiyama, M. et al. Long-acting calcium channel antagonist pranidipine prevents ventricular remodeling after myocardial infarction in rats. Heart Vessels 14, 111–119 (1999). https://doi.org/10.1007/BF02482294
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02482294