Abstract
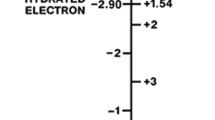
Previous papers by F. M. Snell (Jour. Theor. Biol.,8, 469–479, 1965) and M. A. Fox and H. D. Landahl (Bull. Math. Biophysics,27, Spec. Issue, 183–190, 1965) have found that the formulation by previous authors for the oxygen flow rates through hemoglobin solution as a function of pressure determined by E. Hemmingsen and P. F. Scholander (Science,132, 1379–1381, 1960) did not give a satisfactory quantitative fit of the curve for constant pressure difference. The suggestion of Fox and Landahl that the Bohr effect involving the shift in acidity accompanying the oxidation of Hb should give rise to voltage and pH differences in oxyhemoglobin transport is examined in more detail. In this paper, the previous expressions for the total oxygen flow rate in terms of the end point concentrations are extended to include the effects of the electrical field. Estimates of the potential difference shows it to be negligible. A derivation of a voltage-pH relation shows that the Nernst relation does not apply and a negligible voltage difference does not preclude a pH shift which is the more probable explanation of the discrepancies observed. Several other predictions suitable for experimental testing are made.
Similar content being viewed by others
Literature
Antonini, Eraldo. 1965. “Interrelationship Between Structure and Function in Hemoglobin and Myoglobin.”Physiol. Rev.,45, 123–170.
Collins, R. E. 1961a. “Transport of Gases Through Hemoglobin Solution.”Science,133, 1593–1594.
Collins, R. E. 1961b. “Transport of Gases Through Hemoglobin Solution.”Bull. Math. Biophysics,23, 223–232.
Enns, T. 1964. “Molecular Collision-Exchange Transport of Oxygen by Hemoglobin.”Proc. Nat. Acad. Sci.,51, 247–252.
Fatt, I. and P. F. Scholander. 1960. “Oxygen Diffusion.”Science,132, 368.
— and R. C. La Force. 1961. “Theory of Oxygen Transport Through Hemoglobin solutions.”Science,133, 1919–1921.
Ferry, R. M. and A. A. Green. 1929. “Studies of the Chemistry of Hemoglobin. III. The Equilibrium between Oxygen and Hemoglobin and its Relation to Changing Hydrogen Ion Activity.”Jour. Biol. Chem.,81, 175.
Fox, M. A. and H. D. Landahl. 1965. “Theory of Hemoglobin Facilitated Oxygen Transport.”Bull. Math. Biophysics,27, Spec. Iss., 183–190.
German, B. and J. Wyman, Jr. 1937. “The Titration Curves of Oxygenated and Reduced Hemoglobin.”Jour. Biol. Chem.,117, 533–550.
Gibson, Q. H. 1959. “The Kinetics of Reactions between Haemoglobin and Gases.”Prog. in Biophysics and Biophysical Chem.,9, 2–53.
Hammel, H. T. 1966. “Oxygen Transport through Hemoglobin Solutions.” AMRL-TR-66-19. Wright-Patterson Air Force Base, Ohio.
Haurowitz, F. 1951. “Haemoglobin, Anhydrohemoglobin and Oxyhemoglobin.”Jour. Biol. Chem.,193, 443–452.
Helfferich, F. 1962.Ion Exchange. New York: McGraw-Hill.
Hemmingsen, E. 1962. “Accelerated Exchange of Oxygen-18 Through a Membrane Containing Oxygen-Saturated Hemoglobin.”Science,135, 733–734.
— and P. F. Scholander. 1960. “Specific Transport of Oxygen Through Hemoglobin Solutions.”Science,132, 1379–1381.
— 1965. “Accelerated Transfer of Oxygen Through Solutions of Heme Pigments.”Acta Physiol. Scand.,64 Suppl.246, 1–53.
Hodgman, C. D., ed.Handbook of Chemistry and Physics. 1954–5. 36th Edition. Cleveland: Chemical Rubber Publishing Co.
Keller, K. H. and S. K. Friedlander. 1966. “The Steady State Transport through Hemoglobin Solutions.”Jour. Gen. Physiol.,49, 663–679.
La Force, R. C. 1966. “Steady-State Diffusion in the Carbon-Monoxide Oxygen Hemoglobin System.”Trans. Far. Soc.,62, 1458–1468.
Moore, W. J. 1962.Physical Chemistry. 3rd Edition. Englewood Cliffs, N.J.: Prentice-Hall, Inc.
Pauling, L., H. A. Itano, S. J. Singer, and I. C. Wells. 1949. “Sickle Cell Anemia, A Molecular Disease.”Science,110, 543–548.
Roughton, F. J. W. 1959. “Diffusion and Simultaneous Chemical Reaction Velocity in Haemoglobin Solutions and Red Cell Suspensions.”Prog. in Biophysics and Biophysical Chem.,9, 55–106.
Ruch, Theodore C. and John F. Fulton. 1960.Medical Physiology and Biophysics. Philadelphia: Saunders Co.
Scholander, P. F. 1960. “Oxygen Transport Through Hemoglobin Solutions.”Science,131, 585–590.
— 1965. “Tension Gradients Accompanying Accelerated Oxygen Transport in a Membrane.”Science,149, 876–877.
Snell, Fred M. 1965. “Facilitated Transport of Oxygen Through Solutions of Hemoglobin.”Jour. Theor. Biol. 8, 469–479.
Van Slyke, D. D., R. T. Dillon, and R. Margaria. 1934. “Studies of Gas and Electrolyte Equilibria in Blood. XVIII Solubility and Physical State of Atmospheric Nitrogen in Blood Cells and Plasma.”Jour. Biol. Chem.,105, 571–586.
Wang, J. H. 1961. “Transport of Oxygen Through Hemoglobin Solutions.”Science,133, 1770–1771.
— 1963. “Chemical Reaction Accelerated Transport of Molecules Through Membranes.”Jour. Theor. Biol.,4, 175–178.
Wittenberg, J. B. 1966. “The Molecular Mechanism of Hemoglobin-facilitated Oxygen Diffusion.”Jour. Biol. Chem.,241, 104–114.
Wyman, J., Jr. 1948. “Heme Proteins.”Adv. in Prot. Chem.,4, 407–531.
— 1966. “Facilitated Diffusion and the Possible Role of Myoglobin as a Transport Mechanism.”Jour Biol. Chem.,241, 115–121.
— and D. W. Allen. 1951. “The Problem of the Heme Interactions in Hemoglobin and the Basis of the Bohr Effect.”Jour. Polymer Science,7, 499–518.
Zilversmit, D. B. 1965. “Oxygen-Hemoglobin System: A Model for Facilitated Membranous Transport.”Science,149, 874–876.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bright, P.B. The basic flow equations of electrophysiology in the presence of chemical reactions: II. A practical application concerning the pH and voltage effects accompanying the diffusion of O2 through hemoglobin solution. Bulletin of Mathematical Biophysics 29, 123–138 (1967). https://doi.org/10.1007/BF02476966
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02476966