Abstract
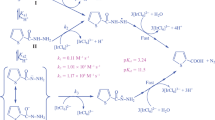
The kinetics of oxidation of diazepam (DZ) by 1-chlorobenzotriazole (CBT) has been studied in HClO4 medium in the temperature range of 293–323 K. The reactions followed a first order dependence on [CBT], fractional order each in [H+] and [DZ]. Activation parameters were evaluated. A negative entropy of activation indicated the involvement of a rigid complex in the activated state. Variation of reaction rate with dielectric constant and the ionic strength are consistent with a mechanism involving the decomposition of a dipole in the rate-determining step. Rate equation is derived to account for the observed kinetic data and the proposed reaction mechanism.
Similar content being viewed by others
References
M. Bedair, M.A. Korany, M.E. Abdul Hamid:Analyst,109, 1423 (1984).
R.C. Hiremath, S.M. Mayanna, N. Venkatasubramanian:J. Sci. and Ind. Res.,49, 122 (1990).
K.V. Uma, S.M. Mayanna:Int. J. Chem. Kinet.,12, 861 (1980).
R.C. Hiremath, S.M. Mayanna, N. VenkatasubramanianJ. Chem. Soc Perkin II, 1569, (1987).
C. Chennegowda, S.M. Mayanna:Ciencia e Cultura,44, 46 (1992).
C.R. Johnson, C.C. Bacon, W.D. Kingsbury:Tetrahedron Lett., 501 (1972).
B. Jayaram, S.M. Mayanna:Tetrahedron,39, 2275 (1983).
C. Chennegowda: Ph.D. Thesis, Bangalore University, India, Jan. 1992, p. 97.
Vogel’sText Book of Quantitative Chemical Analysis, 5th Edition, p.679, ELBS, Longman, 1989.
B.D. Hughes, C.K. Ingold:Quart. Rev.,6, 34 (1952).
N.S. Srinivasan, N. Venkatasubramanian:Tetrahedron,30, 419 (1974).
P.B.D. de la Mare:Electrophilic Halogenation, p. 107. Cambridge University Press, London, 1976.
M.M. Campbell, G. Johnston:Chem. Rev.,78, 65 (1978).
P. Irina, B. Paraschiva, M. Macovschi:Farmacia,25, 241 (1977).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nanda, N., Sheshadri, B.S. & Mayanna, S.M. Mechanism of oxidation of diazepam by 1-chlorobenzotriazole in acidic medium. A kinetic approach. React Kinet Catal Lett 67, 35–41 (1999). https://doi.org/10.1007/BF02475824
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02475824