Abstract
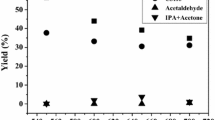
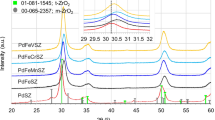
The oxidative dehydrogenation of ethane into ethylene has been investigated on metal oxide-based sulfated zirconia catalysts at temperatures of 400–600°C. It is found that the activity and selectivity toward ethylene depend on the nature of metal oxide and temperature and that Ni and V oxides supported on sulfated zirconia exhibited higher ethylene yields.
Article PDF
Similar content being viewed by others
References
E. Morales, H.H. Lunsford:J. Catal.,118, 255 (1989).
A. Erdõhelyi, F. Solymosi:J. Catal.,123, 31 (1990).
E.M. Kennedy, N.W. Cant:Appl. Catal.,75, 321 (1991).
X. Song, A. Sayari:Catal. Rev.,38, 329 (1996).
T-K. Cheng, B.C. Gates:J. Catal.,168, 522 (1997).
F. Cavani, F. Trifiro:Catal. Today,24, 307 (1995).
C.R. Vera, J.C. Yori, J.M. Parera:Appl. Catal. A.,167, 75 (1998).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wang, S., Murata, K., Hayakawa, T. et al. Oxidative dehydrogenation of ethane over sufated zirconia supported oxides catalysts. React Kinet Catal Lett 67, 219–224 (1999). https://doi.org/10.1007/BF02475763
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02475763