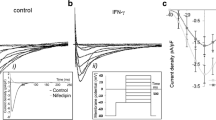
Abstract
Local voltage clamping was applied to mouse macrophage plasma membrane to study calcium channels activated by inositol-1,4,5-triphosphate (IP3) and blocked by heparin. These channels were clearly distinguished from IP3-activated channels of the endoplasmic reticulum by their low conductivity (about 1 pSm for 100 mM Ca2+), high selectivity for Ca2+ relative to K+ (PCa:PK>1000), calcium inactivation, and activation on hyperpolarization; these properties allowed them to be assigned to the ICRAC family. On the other hand, the properties of the IP3 receptors of these channels (IP3R), i.e., the dose-dependent effect of IP3, the IP3 desensitization of the receptor, and the sensitivity to micromolar concentrations of heparin and arachidonic acid were close to those of the endoplasmic reticulum IP3 receptor. The most likely intepretation of these data is that IP3R are not located in the endoplsmic reticulum, but, acting via some kind of conformational change occurring on binding of IP3, transmit a signal from the endoplasmic reticulum to the highly selective Ca2+ channels. This point of view is in agreement with the published “coupling model” [1].
Similar content being viewed by others
References
M. J. Berridge, “Capacitative calcium entry,”Biochem. J.,312, 1–11 (1995).
A. Fabiato and F. Fabiato, “Calculator programs for computing the composition of the solutions containing multiple metals and ligands used for experiments in skinned muscle cells,”J. Physiol. Paris,75, No. 5, 463–505 (1979).
G. Hajnoczky and A. P. Thomas, “The mositol trisphosphate calcium channel is inactivated by inositol trisphosphate,”Nature,370, 474–477 (1994).
B. Hille, in:Ionic Channels of Excitable Membranes, M. A. Sinauer Associates, Sunderland (1992).
M. Hoth and R. Penner, “Calcium release-activated calcium curent in rat mast cells,”J. Physiol. (London),465, 359–386 (1993).
A. A. Khan, J. P. Steiner, M. G. Klein, et al., “IP3 receptor: localization to plasma membrane of T cells and cocapping with the T cell receptor,”Science,257, 815–818.
A. A. Khan, J. P. Steiner, and S. H. Snyder, “Plasma membrane inositol-1,4,5-trisphosphate receptor at lymphocytes: Selective enrichment in the sialic acid and unique binding specificity,”Proc. Natl. Acad. Sci. USA,89, 2849–2853 (1992).
K. I. Kiselyov, A. G. Mamin, S. B. Semyonova, et al., “Lowconductance high selective inositol (1,4,5)-trisphosphate activated Ca2+ channels in plasma membrane of A431 cells,’FEBS Lett.,407, 309–312 (1997).
M. Kuno and P. Gardner, “Ion channels activted by inositol-1,4,5-trisphosphate in human T-lymphocytes,”Nature,326, 301–304 (1987).
A. Luckhoff and D. E. Clapham, “Calcium channels activated by depletion of internal calcium stores in A431 cells,”Biophys. J.,67, 177–182 (1994).
Y. Maruyama, “Control of inositol polyphosphate-mediated calcium mobilization by arachidonic acid in pancreatic acinar cells of rat,”J. Physiol. (London),463, 729–746 (1993).
G. N. Mozhayeva, A. P. Naumov, and Y. A. Kuryshev, “Activation of Ca2+-sensitive potassium channels in A431 cells by epidermal growth factor and histamine,”Biologischeskie Membrany,6, 629–636 (1989).
G. N. Mozhayeva, A. P. Naumov, and Y. A. Kuryshev, “Inositol 1,4,5-trisphosphate activates two types of calcium permeable channels in human carcinoma cells,”FEBS Lett.,277, 233–234 (1990).
J. B. Parys and I. Bezprozyanny, “The inositol trisphosphate receptor ofXenopus oocytes,”Cell Calcium,19, 353–363 (1995).
B. A. Premack, T. V. McDonald, and P. Gardner, “Activation of Ca2+ current in Jurkat T cells following the depletion of Ca2+ stores by microsomal Ca2+-ATP-ase inhibitors,”J. Immunol.,152, 5226–5240 (1994).
J. W. J. Putney, “Capacitative calcium entry revisited,”Cell Calcium,11, 611–624 (1990).
J. W. J. Putney and G. S. J. Birg, “The inositol phosphatecalcium signalling system in nonexcitable cells,”Endocr. Rev.,14, 610–631 (1993).
C. A. Ross, J. Meldolewsi, T. A. Milner, et al., “Inositol 1,4,5-trisphophate receptor localized to endoplasmic reticulum in cerebellar Purkinje neurons,”Nature,339, 468–470 (1989).
S. Rowles and D. V. Gallacher, “Ins(1,3,4,5)P4 is effective in mobilizing Ca2+ in mouse exocrine pancreatic acinar cells in which phospholipase A2 is inhibited,”Biochem. J.,319, 913–918 (1996).
A. Ruchundin, M. J. Song, and F. Sachs, “The ultrastructure of patch-clamped membranes: a study using high voltage electron microscopy,”J. Cell Biol.,112, 125–134 (1991).
L. Vaca and D. L. Kunze, “Deletion of intracellular Ca2+ stores activates a Ca2+-selective channel in vascular endothelium,”Am. J. Physiol.,267, C920-C925 (1994).
L. Vaca and D. L. Kunze, “IP3-activated Ca2+ channels in the plasma membrane of cultured vascular endothelial cells,”Am. J. Physiol.,269, C733-C738 (1995).
A. Zweifach and R. S. Lewis, “Mitogen-regulated Ca2+ current of T lymphocytes is activated by depletion of intracellular Ca2+ stores,”Proc. Natl. Acad. Sci. USA,90, 6295–6299 (1993).
Author information
Authors and Affiliations
Additional information
Translated from Rosiiskii Fiziologicheskii Zhurnal imeni I. M. Sechenova, Vol. 84, No. 5-6, pp. 417–425, May–June, 1998.
Rights and permissions
About this article
Cite this article
Semenova, S.B., Kiselev, K.I. & Mozhaeva, G.N. Low-conductivity calcium channels in the macrophage plasma membrane: Activation by inositol-1,4,5-triphosphate. Neurosci Behav Physiol 29, 339–345 (1999). https://doi.org/10.1007/BF02465347
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02465347