Abstract
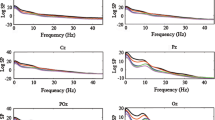
This study was undertaken with the aim of identifying frequency bands with correlated changes in the spectral power amplitudes of brain electrical activity, including high-frequency components (the 1–200 Hz band) in four dogs, using one-dimensional analysis. Factor and cluster analysis of the spectral densities of various parts of the cortex and the olfactory bulb were carried out. The ratios of factors in different parts of the brain, both in terms of the proportions of the total dispersity and in terms of weightings, provided data on regional and individual differences in electrical activity. During learning (development of a motor habit consisting of pressing a feeder pedal), the factor organization of electrical activity became more complex, particularly in the high-frequency part of the spectrum (40–170 Hz). The changes consisted of the appearance of narrower frequency sub-bands, each of which was present at high weighting (0.7–0.9) for one of the factors. The use of high-frequency components allowed functional mosaicism of the neocortex to be detected.
Similar content being viewed by others
References
O. A. Adrianov and T. A. Mering, Atlas of the Dog Brain [in Russian], Medgiz, Moscow (1959).
A. Afifi and S. Eisen, Statistical Analysis. A Computer Approach [Russian translation], Mir, Moscow (1982).
V. N. Dumenko, Learning and the High-Frequency Components of Brain Electrical Activity [in Russian], Nauka, Moscow (1992).
V. N. Dumenko, M. K. Kozlov, and M. A. Kulikov, “Attempts to discriminate the high-frequency range (40–200 Hz) of brain electrical activity in the dog into frequency bands,” Zh. Vyssh. Nerv. Deyat.,45, No. 1, 107 (1995).
V. N. Dumenko, M. A. Kulikov, and M. K. Kozlov, “Is the gamma band of brain electrical activity homogeneous?,” Dokl. Akad. Nauk,339, No. 1, 120 (1994).
K. Iberla, Factor Analysis [in Russian], Statistika, Moscow (1980).
M. K. Kozlov, “Construction of a density profile of the one-dimensional distribution as a cluster approach to the problem of EEG classification,” Zh. Vyssh. Nerv. Deyat.,44, No. 1, 175 (1994).
M. K. Kozlov, “A version of cluster analysis which eliminates the cluster as a vague concept,” Dokl. Akad. Nauk,348, No. 1, 34 (1996).
M. N. Livanov, The Spatial Organization of Brain Processes [in Russian], Nauka, Moscow (1972).
G. Khardman, Current Factor Analysis [in Russian], Statistika, Moscow (1972).
S. L. Bressler, “Relation of olfactory bulb and cortex,” Brain Res.,409, No. 2, 285 (1987).
S. L. Bressler and W. Y. Freeman, “Frequency analysis of olfactory system EEG in cat, rabbit and rat,” EEG Clin. Neurophysiol.,50, No. 1, 19 (1980).
W. Y. Freeman and W. Schneider, “Changes in spatial patterns of rabbit olfactory EEG with conditioning to odors,” Psychophysiol.,19, No. 1, 44 (1982).
Additional information
Institute of Higher Nervous Activity, Russian Academy of Sciences, Moscow. Translated from Zhurnal Vysshei Nervnoi Deyatel'nosti, Vol. 46, No. 4, pp. 719–731, July–August, 195.
Rights and permissions
About this article
Cite this article
Dumenko, V.N., Kozlov, M.K. & Kulikov, M.A. Dynamics of high-frequency (up to 200 Hz) components of brain electrical activity during learning reflect the functional mosaicism of the neocortex. Neurosci Behav Physiol 27, 381–390 (1997). https://doi.org/10.1007/BF02462939
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02462939