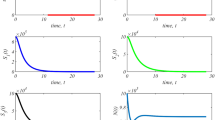
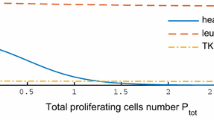
Abstract
A system of differential equations for the control of tumor growth cells in a cycle nonspecific chemotherapy is analyzed. Spontaneously acquired drug resistance is taken into account, and a criterion for the selection of chemotherapeutic treatment is used. This criterion purports to describe the possibility of improvement of the patient's health when treatment is discontinued. Contrary to our early results which also take drug resistance into account, in this context strategies of continuous chemotherapy in which rest periods take part may be better than maximum drug concentration throughout the treatment (which appears to be in accordance with clinical practice). This bears out our previous conjecture that when drug resistance is accounted for, the imperfections in the usual modelling of treatment criteria, which in general do not allow for patient recuperation, ruled out the possibility of rest periods in optimal continuous chemotherapy.
Similar content being viewed by others
References
Bellomo, N. and G. Forni. 1994. Dynamics of tumor interaction with the host immune system.Math. Comput. Modelling 20, 107–122.
Bernardo Strada, M. R., G. Bernardo and G. Robustelli Della Cuna. 1983. Principi di immunoterapia antitumorale. InManuale de Oncologia Medica, G. Bonadonna (Ed.) Paris: Masson.
Clarke, F. H. 1983.Optimization and Nonsmooth Analysis, New York: Wiley.
Coldman, A. J. and J. H. Goldie. 1983. A model for the resistance of tumor cells to cancer chemotherapeutic agents.Math. Biosci. 65, 291–307.
Coldman, A. J. and J. H. Goldie. 1986. A stochastic model for the origin and treatment of tumors containing drug-resistant cells.Bull. Math. Biol. 48, 279–292.
Costa, M. I. S., J. L. Boldrini and R. C. Bassanezi. 1992. Optimal chemical control of populations developing drug resistance.IMA J. Math. Appl. Med. Biol. 9, 215–226.
Costa, M. I. S., J. L. Boldrini and R. C. Bassanezi. 1994. Optimal chemotherapy: a case study with drug resistance, saturation effect and toxicity.IMA J. Math. Appl. Med. Biol. 11, 45–59.
Costa, M. I. S., J. L. Boldrini and R. C. Bassanezi. 1995a. Drug kinetics and drug resistance in optimal chemotherapy.Math. Biosci. 125, 191–209.
Costa, M. I. S., J. L. Boldrini and R. C. Bassanezi. 1995b. Chemotherapeutic treatments involving drug resistance and level of normal cells as a criterion of toxicity.Math. Biosci. 125, 211–228
Eisen, M. 1978.Mathematical Models in Cell Biology and Cancer Chemotherapy. Lecture Notes in Biomathematics, Vol. 30. New York: Springer-Verlag.
Goldie, J. H. and A. J. Coldman. 1979. A mathematical model for relating the drug sensitivity of tumors to their spontaneous mutation rate.Cancer Treat. Rep. 63, 1727–1733.
Harnevo, L. and Z. Agur. 1992. Drug resistance as a dynamic process in a model for multistep gene amplification under various levels of selection stringency.Cancer Chemother. Pharmacol. 30, 469–476.
Herson, J. 1980. Evaluation of toxicity: statistical considerations.Cancer Treat. Rep. 64, 463–468.
Kimmel, M. and D. E. Axelrod. 1990. Mathematical models for gene amplification with application to cellular drug resistance and tumorigenicity.Genetics 125, 633–644.
Kimmel, M., D. E. Axelrod and G. M. Wahl. 1992. A branching process model of gene amplification following chromosome breakage.Mut. Res. 276, 225–239.
Kuznetsov, V. A., I. A. Makalin, M. A. Taylor and A. S. Perelson. 1994. Nonlinear dynamics of immunogenic tumors: parameter estimation and global bifurcation analysis.Bull. Math. Biol. 56, 295–321.
Marusic, M., Z. Bajzer, S. Vuk-Pavlovic and J. P. Fryer. 1994. Tumor growthin vivo and as multicellular spheroids compared by mathematical models.Bull. Math. Biol. 56, 617–631.
Mohler, R. R., K. S. Lee, A. L. Asachenkov and G. I. Marchuk. 1994. A system approach to immunology and cancer.IEEE Trans. Syst. Cybernetics 24, 632–641.
Murray, J. M. 1990a. Optimal control for a cancer chemotherapy problem with general growth and loss functions.Math. Biosci. 98, 273–287.
Murray, J. M. 1990b. Some optimal control problems in cancer chemotherapy with a toxicity limit.Math. Biosci. 100, 49–67.
Murray, J. M. 1995. An example of the effects of drug resistance on the optimal schedule for a single drug in cancer chemotherapy.IMA J. Math. Appl. Med. Biol. 12, 55–71.
Schandl, F. R. 1989. Optimal treatment strategies in cancer chemotherapy. Forschungsbericht Nr. 116, Institut für Ökonometrie, OR und Systemtheorie, Technische Universität Wien.
Skipper, H. E. 1983. The forty year old mutation theory of Luria and Delbruck and its pertinence to cancer chemotherapy.Adv. Cancer Res. 40, 331.
Swan, G. W. 1987. Tumor growth models and cancer chemotherapy. InCancer Modeling, J. R. Thompson and B. W. Brown (Eds). New York: Dekker.
Swan, G. W. 1990. Role of optimal control theory in cancer chemotherapy.Math. Biosci. 101, 237–284.
Swan, G. W. and T. L. Vincent. 1977. Optimal control analysis in the chemotherapy of IgG multiple myeloma.Bull. Math. Biol. 39, 317.
Thornton, B. S. 1988. Prescheduling graphic displays for optimal cancer therapies to reveal possible tumor regression or stabilization.J. Med. Syst. 12, 31–41.
Vaidya, V. G. and F. J. Alexandro, Jr. 1982. Evaluation of some mathematical models for tumor growth.Int. J. Bio-Med. Comp. 13, 19–35.
Vendite, L. L. 1988. Modelagem matemática para o crescimento tumoral e o problema de resistência celular aos fármacos anti-bláticos. Ph.D. thesis, Faculdade de Engenharia Elétrica, Universidade Estadual de Campinas, SP, Brazil.
Vietti, T. J. 1980. Evaluation of toxicity: clinical issues.Cancer Treat. Rep. 64, 457–461.
Zietz, S. and C. Nicolini. 1979. Mathematical approaches to optimization of cancer chemotherapy.Bull. Math. Biol. 41, 305–324.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Costa, M.I.S., Boldrini, J.L. Chemotherapeutic treatments: A study of the interplay among drug resistance, toxicity and recuperation from side effects. Bltn Mathcal Biology 59, 205–232 (1997). https://doi.org/10.1007/BF02462001
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02462001