Abstract
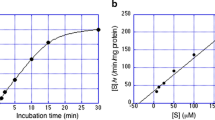
On repeated thawing at room temperature of frozen preparations of heavy microsomes from rat livers, HMGCoA reductase activity was solubilized due to limited proteolysis. This soluble enzyme was partially purified by fractionation with ammonium sulfate and filtration on Sephacryl S-200 column. The active enzyme was coeluted with a major 92 kDa-protein and was identified as a 58kDa-protein after separation by SDS-PAGE and immunoblotting. Ethoxysilatrane, a hypocholesterolemic compound, which decreased the liver-microsomal activity of HMGCoA reductase on intra-peritonial treatment of animals, showed little effect on the enzyme activity with isolated microsomes or the 50kDa-soluble enzyme when added in the assay. But it was able to inhibit the activity of the soluble 58kDa-enzyme in a concentration-dependent, reversible manner. Cholesterol and an oxycholesterol were without effect whereas chlorophenoxyisobutyrate and ubiquinone showed small inhibition under these conditions. The extra region that links the active site domain (50kDa protein) to the membrane, present in the 58kDa-protein appears to be involved in mediating the inhibition by silatrane.
Similar content being viewed by others
References
Gould RG, Taylor B, Hagerman JS, Warner I, Campbell DJ: Cholesterol metabolism. I. Effect of dietary cholesterol on the synthesis of cholesterol in dog tissuein vitro. J Biol Chem 201: 519–528, 1953
Tomkins GM, Sheppard H, Chaikoff IL: Cholesterol synthesis by liver. III. Its regulation by ingested cholesterol. J Biol Chem 201: 137–141, 1953
Bucher NLR, Overath P, Lynen F: 3-Hydroxy-3-methylglutaryl coenzyme A reductase: cleavage and condensing enzymes in relation to cholesterol formation in rat liver. Biochim Biophys Acta 40: 491–501, 1960
Higgins MJP, Rudney H: Regulation of rat liver 3-hydroxy-3-methylglutaryl-CoA reductase activity by cholesterol. Nature New Biol 246: 60–81, 1973
Goldstein JL, Brown MS: Regulation of the mevalonate pathway. Nature (Lond) 343: 425–430, 1990
Thorp JM, Waring WS: Modification of metabolism and distribution of lipids by ethyl chlorophenoxyisobutyrate. Nature (Lond) 194: 948–949, 1962
Avoy DR, Swyryd EA, Gould RG: Effects of α-p-chlorophenoxyisobutyryl ethyl ester (CPIB) with and without androsterone on cholesterol biosynthesis. J Lipid Res 6: 369–376, 1965
Krishnaiah KV, Inamdar AR, Ramasarma T: Regulation of steroidogenesis by ubiquinone. Biochim Biophys Res Communs 27: 474–478, 1976
Krishnaiah KV, Ramasarma T: Regulation of hepatic cholesterolgenesis by ubiquinone. Biochim Biophys Acta 202: 332–342, 1970
Nair N, Kurup CKR: Investigations on the mechanism of the hypocholesterolemic action of diethylhexyl phthalate in rats. Biochem Pharmacol 35: 3441–3447, 1986
Mehta PP, Ramasarma T, Ramakrishna Kurup CK: Hypocholesterolemic action of 1-ethoxysilatrane. Biochim Biophys Acta 920: 102–104, 1987
Mehta PP, Ramasarma T, Ramakrishna Kurup CK: Investigations on the mechanism of the hypocholesterolemic action of 1-ethoxysilatrane. Mol Cell Biochem 97: 75–85, 1990
Kawachi T, Rudeny H: Solubilization and purification of 3-hydroxy-3-methylglutaryl coenzyme A reductase from rat liver. Biochemistry 9: 1700–1705, 1970
Gil G, Faust JR, Chin DJ, Goldstein JL, Brown MS: Membrane-bound domain of HMGCoA reductase is required for sterolenhanced degradation of the enzyme. Cell 41: 249–258, 1985
Heller RA, Gould RG: Solubilization and partial purification of hepatic 3-hydroxy-3-methylglutaryl coenzyme A reductase. Biochem Biophys Res Communs 50: 859–865, 1973
Ness GC, Way SC, Wickham PD: Proteinase involvement in the solubilization of 3-hydroxy-3-methylglutaryl coenzyme A reductase. Biochem Biophys Res Communs 102: 81–85, 1981
Srikantaiah MV, Tormanen CD, Redd WL, Hardgrave JE, Scallen TJ: Purification of 3-hydroxy-3-methylglutaryl coenzyme A reductase by affinity chromatography on blue dextran/sepharose 4B. J Biol Chem 252: 6145–6150, 1977
Ness GC, Spindler CD, Moffler MH: Purification of 3-hydroxy-3-methylglutaryl coenzyme A reductase from rat liver. Arch Biochem Biophys 197: 493–499, 1979
Liscum L, Cummings RD, Anderson RGW, De Martin GN, Goldstein JL, Brown MS: 3-hydroxy-3-methylglutaryl CoA reductase, a transmembrane glyco-protein of the endoplasmic reticulum with N-linked ‘high mannose’ oligosaccharides. Proc Natl Acad Sci (USA) 80: 7165–7169, 1983
Shapiro DJ, Nordstrom JL, Mitschelen JJ, Rodwell VW, Schimke RJ: Microassay for 3-hydroxy-3-methylglutaryl coenzyme A reductase and cholesterol 7-hydroxylase. Biochim Biophys Acta 370: 369–377, 1969
Omkumar RV, Banerji A, Ramakrishna Kurup CK, Ramasarma T: The nature of inhibition of 3-hydroxy-3-methylglutaryl CoA reductase by garlic-derived diallyl disulfide. Biochim Biophys Acta 1078: 219–225, 1991
Gornall AG, Bardawill CJ, David MM: Determination of serum proteins by means of biuret reaction. J Biol Chem 177: 751–766, 1949
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ: Protein measurements with Folin phenol reagent. J Biol Chem 193: 265–275, 1951
Laemmli UK: Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature (Lond) 227: 680–685, 1970
Towbin H, Staehelin T, Gordon T: Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. Proc Natl Acad Sci (USA) 76: 4350–4354, 1979
Liscum L, Finer-Moore J, Stroud RM, Luskey KL, Brown MS, Goldstein JL: Domain structure of 3-hydroxy-3-methylglutaryl coenzyme A reductase, a glycoprotein of the endoplasmic reticulum. J Biol Chem 260: 522–530, 1985
Parker RH, Miller SJ, Gibson DM: Phosphorylation of native 97-kDa 3-hydroxy-3-methylglutaryl-coenzyme A reductase from rat liver. J Biol Chem 264: 4877–4887, 1989
Mayer RJ, Debouck C, Metcalf BW: Purification and properties of the catalytic domain of human 3-hydroxy-3-methylglutaryl-CoA reductase expressed inEscherichia coli. Arch Biochem Biophys 267: 110–118, 1988
Von Gunter CF, Sinensky M: Treatment of CHO-K1 cells with 25-hydroxycholesterol produces a more rapid loss of HMGR activity than can be accounted for by enzyme turnover. Biochem Biophys Acta 1001: 218–224, 1989
Beg ZH, Allman DW, Gibson DM: Modulation of 3-hydroxy-3-methylglutaryl coenzyme A reductase activity with cAMP and with protein fractions of rat liver cytosol. Biochem Biophys Res Communs 54: 1362–1369, 1973
Kennelly PJ, Rodwell VW: Regulation of 3-hydroxy-3-methylglutaryl coenzyme A reductase by reversible phosphorylation-dephosphorylation. J Lipid Res 26: 903–914, 1985
Ness GC, Benton GA, Deiter SA, Wickham PS: Influence of mevalonate kinase on studies of the MgATP-dependent inactivator of 3-hydroxy-3-methylglutaryl coenzyme A reductase. Arch Biochem Biophys 214: 705–713, 1982
Satish Menon A, Usha Devi S, Ramasarma T: Inhibition of 3-hydroxy-3-methylglutaryl coenzyme A reductase by cytosolic proteins — real or artifact. Biochem Biophys Res Communs 109: 619–625, 1982
Cappel RE, Gilbert HF: Thioldisulfide exchange between 3-hydroxy-3-methylglutaryl coenzyme A reductase and glutathione. J Biol Chem 263: 12204–12212, 1988
Dotan I, Shechter I: Thiol-disulfide-dependent interconversion of active and latent forms of rat hepatic 3-hydroxy-3-methylglutaryl coenzyme A reductase. Biochim Biophys Acta 713: 427–434, 1982
Usha Devi S, Ramasarma T: Hemin-mediated oxidative inactivation of 3-hydroxy-3-methylglutaryl CoA reductase. Mol Cell Biochem 77: 103–111, 1987
Menon AS, Usha Devi S, Ramasarma T: Purification and characterization of fermodulin — an Fe2+-dependent inhibitor protein of 3-hydroxy-3-methylglutaryl CoA reductase. Arch Biochem Biophys 239: 342–352, 1987
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Omkumar, R.V., Mehta, P.P., Ramakrishna Kurup, C.K. et al. Preparation of a soluble 58kDa-3-hydroxy-3-methylglutaryl CoA reductase from liver microsomes and its inhibition by ethoxysilatrane, a hypocholesterolemic compound. Mol Cell Biochem 110, 145–153 (1992). https://doi.org/10.1007/BF02454191
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02454191