Summary
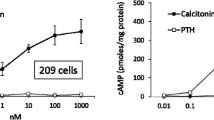
A cell line (LLC-PK1) isolated from porcine kidney increases 3'5'-cyclic adenosine monophosphate (cAMP) content when incubated with salmon calcitonin (SCT) or antidiuretic hormone (ADH). We have examined several factors which modulate the hormone-induced changes in cAMP levels in these cells. Preincubation with increasing concentrations of SCT results in a dose-dependent decrease in cAMP levels in cells retested with this hormone. Addition of 3-isobutyl-1-methylxanthine (IBMX) to cells preincubated with SCT results in 15–30-fold increases in cAMP levels compared to cells preincubated without this hormone. These observations suggest that the decrease in SCT-induced cAMP response in cells pretreated with this hormone is related at least in part to stimulation of phosphodiesterase activity. Preincubation with ADH does not affect subsequent cAMP response to either ADH or SCT, suggesting that these hormones interact with different cell surface receptors. Cell cycle and plating density also affect cAMP levels. cAMP content per cell increases with increasing cell density, which is associated with an increase in the SCT-induced cAMP response. These studies illustrate that factors other than receptor occupancy modulate cAMP responses of these cells to specific hormones.
Similar content being viewed by others
References
Haynes, R.C., Jr.: The activation of adrenal phosphorylase by the adrenocorticotrophic hormone, J. Biol. Chem.233:1220–1222, 1958
Grahame-Smith, D.G., Butcher, R.W., Ney, R.L., Sutherland, E.W.: Adenosine 3′,5′-monophosphate as the intracellular mediator of the action of adrenocorticotrophic hormone on the adrenal cortex, J. Biol. Chem.242:5535–5541, 1967
Birnbaumer, L., Pohl, S.L.: Relation of glucagon-specific binding sites to glucagon-dependent stimulation of adenylylcyclase activity in plasma membranes of rat liver, J. Biol. Chem.248:2056–2061, 1973
Marx, S.J., Fedak, S.A., Aurbach, G.D.: Preparation and characterization of a hormone-responsive renal plasma membrane fraction, J. Biol. Chem.247:6913–6918, 1972
Greengard, P.: Phosphorylated proteins as physiological effectors: protein phosphorylation may be a final common pathway for many biological regulatory agents, Science199:146–152, 1978
Hinkle, P.M., Tashjian, A.H., Jr.: Thyrotropin-releasing hormone regulates the number of its own receptors in the GH3 strain of pituitary cells in culture, Biochemistry14:3845–3851, 1975
Kolata, G.B.: Hormone receptors: how are they regulated? Science196:747–800, 1977
Lefkowitz, R.J., Mullikin, D., Wood, C.L., Gore, T.B., Mukherjee, C.: Regulation of prostaglandin receptors by prostaglandins and guanine nucleotides in frog erythrocytes, J. Biol. Chem.252:5295–5303, 1977
Mickey, J., Tate, R., Lefkowitz, R.J.: Subsensitivity of adenylate cyclase and decreased beta-adrenergic receptor binding after chronic exposure to (minus)-isoproterenol in vitro, J. Biol. Chem.250:5727–5729, 1975
Mukherjee, C., Caron, M.G., Lefkowitz, R.J.: Catecholamine-induced subsensitivity of adenylate cyclase associated with loss of beta-adrenergic receptor binding sites, Proc. Natl. Acad. Sci. U.S.A.72:1945–1949, 1975
Goldring, S.R., Dayer, J.-M., Ausiello, D.A., Krane, S.M.: A cell strain cultured from porcine kidney increases cyclic AMP content upon exposure to calcitonin or vasopressin, Biochem. Biophys. Res. Commun.83:434–440, 1978
Hull, R.N., Cherry, W.R., Weaver, G.W.: The origin and characteristics of a pig kidney cell strain, LLC-PK, In Vitro12:670–677, 1976
Goldring, S.R., Dayer, J.-M., Russell, R.G.G., Mankin, H.J., Krane, S.M.: Response to hormones of cells cultured from human giant cell tumors of bone, J. Clin. Endocrinol. Metab.46:425–433, 1978
Puck, T.T.: Studies of the life cycle of mammalian cells, Cold Spring Harbor Sym. Quant. Biol.29:167–176, 1964
Shear, M., Insel, P.A., Melman, K.L., Coffino, P.: Agonist-specific refractoriness induced by isoproterenol: studies with mutant cells, J. Biol. Chem.251:7572–7576, 1976
Dickinson, J.R., Graves, M.G., Swoboda, B.E.: Induction of division synchrony inTetrahymena pyriformis by a single hypoxic shock. Its use in elucidating control of the cell cycle by adenosine 3',5'-monophosphate, Eur. J. Biochem.78:83–87, 1977
Seifert, W., Rudland, P.S.: Cyclic nucleotides and growth control in cultured mouse cells: correlation of changes in intracellular 3',5'-cAMP concentration with a specific phase of the cell cycle, Proc. Natl. Acad. Sci. U.S.A.71:4920–4924, 1974
Newcombe, D.S., Ciosek, C.P., Jr., Ishikawa, Y., Fahey, J.V.: Human synoviocytes: activation and desensitization by prostaglandins and l-epinephrine, Proc. Natl. Acad. Sci. U.S.A.72:3124–3128, 1975
Conti, M., Harwood, J.P., Hsueh, A.J.W., Dufau, M.L., Catt, K.J.: Gonadotropin-induced loss of hormone receptors and desensitization of adenylate cyclase in the ovary, J. Biol. Chem.251:7729–7731, 1976
Nemecek, G.M., Ray, K.P., Butcher, R.W.: Inhibition of cyclic nucleotide phosphodiesterase during exposure of WI-38 cells to prostaglandin E1, J. Biol. Chem.254:598–601, 1979
Manganiello, V., Vaughan, M.: An effect of insulin on cyclic adenosine 3',5'-monophosphate phosphodiesterase activity in fat cells, J. Biol. Chem.248:7164–7170, 1973
Loten, E.G., Assimacopoulas-Meannet, F.D., Exton, J.H., Park, C.R.: Stimulation of a low Km phosphodiesterase from liver by insulin and glucagon, J. Biol. Chem.253:746–757, 1978
D'Armieto, M., Johnson, G.S., Pastan, I.: Regulation of adenosine 3′,5′-cyclic monophosphate phosphodiesterase activity in fibroblasts by intracellular concentrations of cyclic adenosine monophosphate, Proc. Natl. Acad. Sci. U.S.A.69:459–462, 1972
Schwartz, J.P., Passonneau, J.V.: Cyclic AMP-mediated induction of the cyclic AMP phosphodiesterase of C-6 glioma cells, Proc. Natl. Acad. Sci. U.S.A.71:3844–3848, 1974
Thompson, W.J., Terasaki, W.L., Epstein, P.M., Strada, S.J.: Assay of cyclic nucleotide phosphodiesterase and resolution of multiple molecular forms of the enzyme, Adv. Cyclic Nucleotide Res.10:69–92, 1979
Moran, J., Hunziker, W., Fischer, J.A.: Calcitonin and calcium ionophores: cyclic AMP responses in cells of a human lymphoid line, Proc. Natl. Acad. Sci. U.S.A.75:3984–3988, 1978
Tashjian, A.H., Jr., Wright, D.R., Ivey, J.L., Pont, A.: Calcitonin binding sites in bone: relationships to biological response and “escape,” Recent Prog. Horm. Res.34:285–334, 1978
Wener, J.A., Gorton, S.J., Raisz, L.G.: Escape from inhibition of resorption in cultures of fetal bone treated with calcitonin and parathyroid hormone, Endocrinology90:752–759, 1972
Cidlowski, J.A., Michaels, G.A.: Alteration in glucocorticoid binding site number during the cell cycle in HeLa cells, Nature266:643–645, 1977
Isersky, C., Metzger, H., Buell, D.N.: Cell cycle-associated changes in receptors for IgE during growth and differentiation of a rat basophilic leukemia cell line, J. Exp. Med.141:1147–1162, 1975
Makman, M.H., Klein, M.I.: Expression of adenylate cyclase, catecholamine receptor, and cyclic adenosine monophosphate-dependent protein kinase in synchronized culture of Chang's liver cells, Proc. Natl. Acad. Sci. U.S.A.69:456–458, 1972
Penit, J., Cantau, B., Huot, J., Jard, S.: Adenylate cyclase from synchronized neuroblastoma cells: responsiveness to prostaglandin E1, adenosine, and dopamine during the cell cycle, Proc. Natl. Acad. Sci. U.S.A.74:1575–1579, 1977
Sivak, A.: Induction of cell division in BALB/c-3T3 cells by phorbol myristate acetate or bovine serum: effects of inhibitors of cyclic AMP phosphodiesterase and Na+-K+-AT-Pase, In Vitro13:337–343, 1977
Costa, M., Gerner, E.W., Russell, D.H.: Cyclic AMP levels and types I and II cyclic AMP-dependent protein kinase activity in synchronized cells and in quiescent cultures stimulated to proliferate, Biochim. Biophys. Acta538:1–10, 1978
Holley, R.W.: Control of growth of mammalian cells in cell culture, Nature258:487–490, 1975
Singer, F.R., Keutmann, H.T., Neer, R.M., Potts, J.T., Jr., Krane, S.M.: Pharmacological effects of salmon calcitonin in man. In Talmage, R.V., Munson, P.L. (eds.): Calcium, Parathyroid Hormone and the Calcitonins, pp. 89–96. Excerpta Medica, Amsterdam, 1971
Singer, F.R., Krane, S.M.: Skeletal resistance to salmon calcitonin (SCT) in patients with medullary carcinoma of the thyroid (MCT), Proc. 55th Annu. Meet. Am. Endoc. Soc. Abstract No. 271, 1973
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Héron, JF., Dayer, JM., Goldring, S.R. et al. Factors modulating the response of a porcine renal tubular cell line to calcitonin and antidiuretic hormone. Calcif Tissue Int 33, 41–50 (1981). https://doi.org/10.1007/BF02409411
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02409411