Abstract
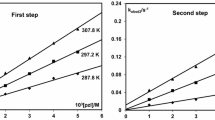
The kinetics of nitration of 2-picrylaminopyridine with nitric acid in 85– 96% sulfuric acid were studied by a spectrophotometric method. It is shown that 2-picrylaminopyridine undergoes the reaction in the protonated form. The kinetic parameters of the nitration and the relative reactivity as compared with benzene were calculated.
Similar content being viewed by others
Literature cited
K. K. Shatemirova, G. A. Davydova, I. V. Verevkina, and A. I. Tochilkin, Vopr. Med. Khim.,23, 609 (1977).
W. Miller, Ber.,23, 2262 (1890).
E. Lellman and H. Alt, Ann.,237. 308 (1887).
G. I. Mikhailov, Tr. IREA,18, 51 (1941).
Ya. I. Gur'yan and Yu. A. Vakhrushev, Zh. Anal. Khim., un16, 485 (1961).
P. B. D. De la Mare, M. Kiamud-din, and J. H. Ridd, J. Chem. Soc., 561 (1960).
H. Gilman and S. M. Spatz, J. Am. Chem. Soc.,63, 1553 (1941).
O. Werner and F. Vögtle, Synthesis,4, 272 (1977).
J. N. Ospenson, Acta Chem. Scand.,4, 1351 (1950).
G. Ganguli, J. Indian Chem. Soc.,11, 197 (1934).
T. Ukai, S. Hashimoto, and M. Higashi, J. Pharm. Soc. Jpn.,48, 371 (1928).
Author information
Authors and Affiliations
Additional information
See [1] for communication 3.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 3, pp. 363–365, March, 1980.
Rights and permissions
About this article
Cite this article
Sharnin, G.P., Falyakhov, I.F. & Khairutdinov, F.G. Reactivities of heterocyclic compounds in niration. 4. Kinetics and mechanism of nitration of 2-picrylaminopyridine in sulfuric acid. Chem Heterocycl Compd 16, 272–274 (1980). https://doi.org/10.1007/BF02401725
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02401725