Abstract
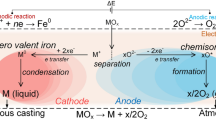
Oxide layers are produced on iron surface, under discharge in water ambient, applying both anodic and cathodic potentials to iron foil. Non-stoichiometric Fe1−xO containing different ratios of Fe3+ and Fe2+ has been detected by using CEMS. Corrosion of these samples in 0.5 M H2SO4 shows that, cathodically discharge treated iron foil has higher corrosion resistance as compared to the anodically treated sample. This is attributed to the microstructural differences in the Fe1−xOx films formed during the discharge.
Similar content being viewed by others
References
V. Ashworth, W.A. Grant, R.P.M. Procter and T.C. Wellington, Corrosion Science16, 393 (1976).
J.W. Mayer and S.S. Lau in Surface Modification and Alloying by Laser, Ion and Electron Beams, edited by J.M. Poate, G. Foti, D.C. Jacobson, (Plenum Press, New York, London, 1981), Chapter 9.
S.B. Ogale, D.M. Phase, S.M. Chaudhari, S.V. Ghaisas, S.M. Kanetkar, P.P. Patil, V.G. Bhide and S.K. Date, Phy. Rev. B.35(4), 1593 (1987).
C.W. Draper and J.M. Poate in. Surface Modification and Alloying by Laser, Ion and Electron Beams, edited by J.M. Poate, G. Foti, D.C. Jacobson, (Plenum Press, New York, 1981), Chapter 13.
K.K. Namitokov, Sov. Phys. (Tech. Phys.)12(5), 714 (1967).
Agnieszka Pattek-Janczyk, Bogdan Sapiol, Jean Claude Greiner & Leopold Fournes, Mat. Res. Bull21(9), 1083 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bulakh, N.R., Ghaisas, S.V., Kulkarni, S.A. et al. Surface oxides produced during discharge in water ambient iron surface: A conversion electron Mössbauer spectroscopic study. Hyperfine Interact 41, 641–644 (1988). https://doi.org/10.1007/BF02400472
Issue Date:
DOI: https://doi.org/10.1007/BF02400472