Abstract
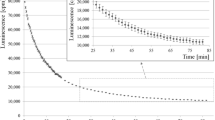
Liquid scintillation counting of40K in ordinary potassium propionate is a highly suitable experimental task in the general education on radioactivity. The counting efficiency is about 90%, depending little on the measurement conditions. Potassium propionate is of suitable properties and can be easily obtained commercially or by conversion from other compounds. The result of counting, about 1,700 cpm/g, is highly impressive to many students, indicating the existence of40K radioactivity unexpectedly high in the normal nature, and can be used for the exercise of calculating our internal radiation exposure.
Similar content being viewed by others
References
C. M. Liderer, V. S. Shirley, Table of Isotopes, 7th ed., John Willey, New York, 1978, p. 90.
W. D. Ehmann, D. E. Vance, Radiochemistry and Nuclear Method of Analysis. John Willey, New York, 1991, p. 179.
J. C. Norma, J. Chem. Educ., 62 (1985) 439.
G. B. Forbes, Body Composition, in: Modern Nutrition in Health and Desease, Vol. 1,M. E. Shils, J. A. Olson andM. Shike (Eds), 8th ed., New York, 1994, p. 781.
Ref. 2,, p. 41.
T. Matsuura, R. Miki, The 33rd Annual Meeting on Radioisotopes in Physical Sciences and Industries, Tokyo, July, 1996, Abstract, p. 23.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ogawa, K., Saito, J. & Nozaki, T. Liquid scintillation measurement of40K for the education on radioactivity. J Radioanal Nucl Chem 239, 657–659 (1999). https://doi.org/10.1007/BF02349086
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02349086