Abstract
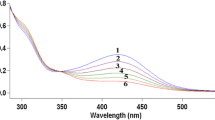
Reactions of carbonate radical (Co3 −) generated by photolysis or by radiolysis of a carbonate solution, with Cu(II) complexes of aminopolycarboxylic acids viz., Cu(II)ethylenediamine tetraacetate [CuIIEDTA]2− and Cu(II)-iminodiacetate [CuIIIDA] were studied at pH 10. 5 and ionic strength 0.2 mol·dm−3. Time-resolved spectroscopy and kinetics for the transients were studied using flash photolysis and stable products arising from the ligand degradation of the complex were ascertained by steady-state radiolysis experiments. From the kinetic data it is observed that CO3 −, radical reacts initially with CuII-complex to form a transient intermediate having maximum absorption at 335 nm and 430 nm. From the subsequent reactions of this intermediate it was assigned to be CuIII. species. This Cu(III) species undergoes intermolecular electron transfer with the CuII-complex to give a radical intermediate which again slowly reacts with CuII-complex to give a long lived species containing Cu−C bond. This long lived species, however, slowly decomposed to give glyoxalic reaction between CuIII-complex and a suitable donor, the one electron reduction potential for [CuIIIEDTA]1−/[CuIIEDTA]2− and [CuIIIIDA]+1/CuIIIDA was determined.
Similar content being viewed by others
References
S. N. Bhattacharyya, K. P. Kundu, Intern. J. Radiat. Phys. Chem., 3 (1971) 1.
S. N. Bhattacharyya, K. P. Kundu, Intern. J. Radiat. Phys. Chem. 4 (1972) 31.
S. N. Bhattacharyya, K. P. Kundu, Radiat. Res., 51 (1972) 45.
S. N. Bhattacharyya, K. P. Kundu, Intern. J. Radiat. Phys. Chem., 5 (1973) 183.
K. P. Kundu, N. Matsuura, Intern. J. Radiat. Phys. Chem., 7 (1975) 565.
J. Lati, D. Meyerstein, Intern. J. Radiat. Phys. Chem., 7 (1975) 611.
S. N. Bhattacharyya, K. P. Kundu, Radiat. Eff., 29 (1976) 231.
S. N. Bhattacharyya, E. V. Srisankar, Intern. J. Radiat. Phys. Chem., 8 (1976) 667.
S. N. Bhattacharyya, N. C. Saha, Radiat. Res., 68 (1976) 234.
S. N. Bhattacharyya, E. V. Srisankar, Radiat. Res., 71 (1977) 325.
S. N. Bhattacharyya, E. V. Srisankar, J. Chem. Soc., Faraday Trans. 1, 74 (1978) 622.
S. N. Bhattacharyya, N. C. Saha, J. Chem. Res. (S), (1978) 70.
J. Lati, D. Meyerstein, J. Chem. Soc., Dalton Trans., (1979) 1105.
E. V. Srisankar, S. N. Bhattacharyya, J. Chem. Soc., Dalton Trans., (1980) 675.
E. V. Srisankar, N. C. Saha, P. C. Mandal, S. N. Bhattacharyya, J. Chem. Soc., Dalton Trans., (1984) 1629.
P. C. Mandal, D. K. Bardhan, S. Sarkar, S. N. Bhattacharyya, J. Chem. Soc., Dalton Trans., (1991) 1457.
P. C. Mandal, D. K. Bardhan, S. N. Bhattacharyya, J. Radioanal. Nucl. Chem., 191 (1995) 349.
S. N. Bhattacharyya, N. C. Saha, Radiat. Effects, 42 (1979) 191.
I. G. Draganic, Z. D. Draganic, The Radiation Chemistry of Water, Academic Press, New York, 1971.
T. E. Friedemann, G. E. Haugen, J. Biol. Chem., 147 (1943) 415.
C. E. Bricker, H. R. Johnson, Ind. Eng. Chem. Anal., 17 (1945) 400.
J. W. T. Spinks, R. J. Woods, An Introduction to Radiation Chemistry, 2nd ed., Wiley, New York, 1976, p. 247.
F. S. Dainton, D. B. Peterson, Nature, 186 (1960) 878.
F. S. Dainton, D. B. Peterson, Proc. R. Soc. London, Ser A, 267 (1962) 443.
M. Anbar, M. Bambenek, A. B. Ross, Nat. Stand. Ref. Data. Ser., Nat. Bur. Stand., 1973, No 43.
G. E. Adams, J. W. Boag, Proc. Chem. Soc., London, (1964) 112.
M. Anbar, P. Neta, Intern. J. Appl. Radiation Isotopes, 18 (1967) 493.
E. Hayon, J. J. McGarvey, J. Phys. Chem., 71 (1967) 1472.
S. Chen, V. W. Cope, M. Z. Hoffman, J. Phys. Chem., 77 (1973) 1111.
S. V. Jovanovic, M. G. Simic, J. Phys. Chem, 90 (1986) 947.
A. Henglein, Radiat. Phys. Chem., 15 (1980) 151.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mandal, P.C., Bardhan, D.K. Oxidation of Cu(II) aminopolycarboxylates by carbonate radical: A flash photolysis study. J Radioanal Nucl Chem 241, 553–560 (1999). https://doi.org/10.1007/BF02347212
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02347212