Abstract
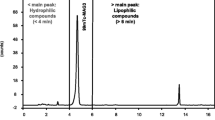
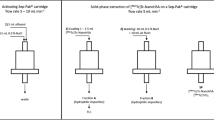
Quality Control (QC) of radiopharmaceuticals is important, providing products of high standards. The current study refers to an enzymic radioiodination method applied to the labeling of antibodies used in nuclear medicine and to the respective QC procedures. Readily available materials such as125I, polyclonal immunoglobulin G (IgG) and a collection of simple chromatographic techniques helped establish a QC protocol suitable for the radiochemical evaluation of radioiodinated biomolecules. A high pre-purification radiolabeling yield (90%) for total protein was detected by paper electrophoresis (PE) and size exclusion chromatography (SEC), but only the inclusion of SDS-PAGE confirmed the existence of “self-iodination” of lactoperoxidase (LPO) and revealed the true radiolabeling yield for125I-IgG (79%).
Similar content being viewed by others
References
Y. Jean-Baptiste, Nucl. Med. Biol., 13 (1986) 221.
Note for Guidance, Pharmacol. and Toxicol., 69 (1991) 218.
D. Skarlos, G. Aravantinos, P. Papakostas, J. Mpeis, A. Varvarigou, A. Epenetos, Arch Hellen. Med., 4 (1987) 138.
J. M. Ferens, K. A. Krohn, P. L. Beaumier, J. P. Brown, I. Hellstrom, K. E. Hellstrom, J. A. Carasquillo, S. M. Larson J. Nucl. Med., 25 (1984) 367.
H. J. Haimsa, J. Hilgers, V. R. Zurawski, J. Nucl. Med., 27 (1986) 1890.
G.S. David, Biochem. Biophys. Res. Comm., 48 (1972) 464.
C. C. Kuenzel, M. Dobeli, Experientia, 29 (1973) 800.
B. L. Wajchenberg, H. Pinto, I. Torres de Toledo e Souza, A. C. Lerario, R. R. Pieroni J. Nucl. Med., 19 (1978) 900.
P. J. Fraker, J. C. Speck, Biochem. Biophys. Res. Comm., 80 (1978) 849.
M. Morrison, Methods Enzymol., 70 (1980) 214.
S. M. Larson, J. Nucl. Med., 26 (1985) 538.
J. C. Saccavini, J. Bohy, J. Bruneau Nucl Med. Biol., 13 (1986) 191.
D. J. Hnatowich, Nucl. Med. Biol., 17 (1990) 49.
C. R. Divgi, A. M. Scott, L. Dantis, P. Capitelli, K. Siler, S. Hilton, R. D. Finn, N. Kemeney, D. Kelsen, L. Kostakoglu, J. Schlom, S. M. Larson, J. Nucl. Med., 36 (1995) 586.
Ch. A. Bizollon, G. Galy Nucl. Med. Biol., 13 (1986) 223.
S. C. Archimandritis, J. Malamitsi, E. Belkas, A. D. Varvarigou, K. K. Cotsyfakis, G. Sivolapenko, J. Zorzos, D. V. Skarlos, C. Proukakis, J. Label. Comp. Radiopharm. 34 (1993) 323.
S. C. Archimandritis, K. Sekeri-Pataryas, G. Sivolapenko, E. Paschali, A. D. Varvarigou, Tumor Targetting, 2 (1996) 85.
A. D. Varvarigou, S. C. Archimandritis, K. Sekeri-Pataryas, T. G. Sourlingas, G. Sivolapenko, E. Paschali, Nucl. Med. Comm., 17 (1996) 80.
P. Angelberguer, I. Virgolini, O. Buchheit, M. Egger, R. Portner, H. Kvaternick, J. Label. Comp. Radiopharm., 37 (1995) 502.
U. K. Laemmli, Nature, 277 (1970) 680.
K. D. Prenzel, S. Thones, B. H. Havsten, Hoppe-Seyler's Z. Physiol. Chem., 358 (1977) 1483.
M. Juweid, J. Sato, C. Paik, S. Onay-Basaran, J. N. Weinstein, R. D. Neumann, Nucl Med. Biol., 20 (1993) 311.
B. K. Seon, D. Pressman, Biochim. Biophys. Acta, 577 (1979) 248.
J. Gow, A. C. Wardlaw, Biochem. Biophys. Res. Comm., 67 (1975) 43.
A. H. Gobuty, E. E. Kim, R. E. Weiner, J. Nucl. Med., 26 (1985) 546.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Potamianos, S., Varvarigou, A.D., Costopoulos, B. et al. Establishing a quality control protocol for the radiochemical evaluation of radioiodinated biomolecules with application in nuclear medicine. J Radioanal Nucl Chem 242, 437–444 (1999). https://doi.org/10.1007/BF02345575
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02345575