Abstract
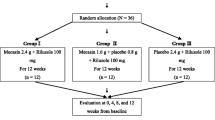
The present study set out to define the possible effect of reduced glutathione (GSH), the substrate of glutathione peroxidase (GSH-Px), a free radical inactivating enzyme, in amyotrophic lateral sclerosis (ALS). Thirty-two patients affected by definite ALS seen in our institution between August 1993 and July 1994 were admitted to the study. The effect of GSH was studied in an open, crossover, randomized study. GSH was given at the dose of 600 mg each day intramuscularly for 12 weeks. The patients, taken sequentially, were randomly assigned to two groups. The first group received the drug while the second received only symptomatic therapies for 12 weeks. After a week of wash-out, the second group received GSH and the first only symptomatic therapies for 12 weeks. The rate of progression of the diseases was compared in the two groups. Clinical evaluation included manual test for muscle strength, Norris scale, bulbar scale, and forced vital capacity (FVC) percent. No significant difference was found in the progression of ALS in the two periods, although a slight slowing of the disease progression rate was found during the period of treatment, probably related to the open design of the study. Our data do not show any significant effect of reduced glutathione in modifying the progression of ALS.
Sommario
Lo scopo del lavoro è stato definire il possibile effetto del glutatione ridotto (GSH), il substrato della glutatione perossidasi (GSH-Px), un enzima inattivatore dei radicali liberi, nella sclerosi laterale amiotrofica (SLA). Sono stati ammessi nello studio 32 pazienti affetti da SLA definita visitati nel nostro Istituto dall'agosto 1993 a luglio 1995. L'effetto del GSH è stato studiato in uno studio in aperto, crossover, randomizzato. Il GSH è stato somministrato alla dose di 600 mg per via intramuscolare per 12 settimane. I pazienti sono stati assegnati a caso, secondo l'ordine di visita, a due gruppi. Per le prime 12 settimane il primo gruppo ha ricevuto il farmaco e il secondo solo terapie sintomatiche. Dopo un wash-out di una settimana, il secondo gruppo ha ricevuto il GSH e il primo solo terapie sintomatiche per altre 12 settimane. È stata confrontata la velocità di progressione dei sintomi nei due gruppi. La valutazione clinica comprendeva test manuali per la forza muscolare, la scala di Norris, una scala bulbare e la capacità vitale forzata (FVC). Non è stata trovata alcuna differenza significativa nella velocità di progressione della SLA nei due periodi, anche se è stato osservato un lieve rallentamento nella velocità di progressione durante il perrodo di trattamento, forse dovuto al disegno in aperto dello studio. I nostri dati non dimostrano alcun effetto significativo del glutatione ridotto nel modificare la progressione della SLA.
Similar content being viewed by others
References
Rosen DR, Siddique T, Patterson D, Figlewicz DA, Sapp P, Hentati A, Donaldson D, Goto J, O'Regan JP, Deng HX (1993) Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature 362(6415):59–62
Smith RG, Appel SH (1995) Molecular approaches to amyotrophic lateral sclerosis. Annu Rev Med 46:133–145
Rothstein JD, Kuncl RW (1995) Neuroprotective strategies in a model of chronic glutamate-mediated motor neuron toxicity. J Neurochem 65:643–651
Sardesai VM (1995) Role of antioxidant in health maintenance. Nutr Clin Pract 10:19–25
Bains JS, Shaw CA (1997) Neurodegenerative disorders in humans: The role of glutathione in oxidative stress-mediated neuronal death. Brain Res Brain Res Rev 25:335–358
Mitchell JD, Jackson MJ (1992) Free radicals, amyotrophic lateral sclerosis, and neurodegenerative disease. In: Smith RA (ed) Handbook of amyotrophic lateral sclerosis. Marcel Dekker, New York, pp 533–54
Moumen R, Nouvelot A, Duval D, Lachevalier B, Viader F (1997) Plasma superoxide dismutase and glutathione peroxidase activity in sporadic amyotrophic lateral sclerosis. J Neurol Sci 151:35–39
World Federation of Neurology Research Group on Neuromuscular Diseases and Subcommittee on Motor Neuron Diseas e/Amyotrophic Lateral Sclerosis (1994) El Escorial World Federation of Neurology criteria for the diagnosis of amyotrophic lateral sclerosis. J Neurol Sci 124 [Suppl]:96–107
The Italian ALS Study Group (1993) Branched-chain amino acids and amyotrophic lateral sclerosis: A treatment failure? Neurology 43:2466–2470
Daniel WW (1978) Biostatistics: A foundation for analysis in the health sciences, 2nd edn. John Wiley & Sons, New York
CPMP Working Party on Efficacy of Medical Products (1990) EEC note for guidance: Good clinical practice for trials on medical products in the European Community. Pharm Toxicol 67:361–372
Swash M, Schwartz MS (1992) What do we really known about amyotrophic lateral sclerosis? J Neurol Sci 113:4–16
Kannan R, Kuhlenkamp JF et al (1990) Evidence for carrier-mediated transport of glutathione across blood-brain barrier in the rat. J Clin Invest 85:2009–2013
Lanius RA, Krieger C, Wagey R, Shaw CA (1993) Increased [35S]glutathione binding sites in spinal cord from patients with sporadic amyotrophic lateral sclerosis. Neurosci Lett 163:89–92
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chili, A., Cucatto, A., Terreni, A.A. et al. Reduced glutathione in amyotrophic lateral sclerosis: an open, crossover, randomized trial. Ital J Neuro Sci 19, 363–366 (1998). https://doi.org/10.1007/BF02341783
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02341783