Summary
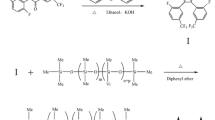
A propyldimethylphenylsilane stationary phase was prepared by a hydrosilylation reaction on the double bond of an allyl bonded phase intermediate. The carbon load on the silica was between 8.8–9.0%. Evaluation of the material by FTIR indicated high conversion of the double bond by the hydrosilylation reaction. The bonded material displayed reversedphase properties as determined by the retention behavior of alkylarylketones but it was less hydrophobic than either C-8 or C-18. The stability at both low and high pH was excellent. Separations of pharmaceutical compounds, a mixture of anilines, and a mixture of dopamine and epinephrine were satisfactory.
Similar content being viewed by others
References
J. J. Pesek, S. A. Swedberg, J. Chromatogr.,361, 83 (1986).
J. J. Pesek, P. Mahabadi, S. J. Chan, Chromatographia,23, 3 (1987).
J. J. Pesek, G. Guiochon, J. Chromatogr.,395, 1 (1987).
J. J. Pesek, W. Y. Wey, Chromatographia,25, 969 (1988).
J. J. Pesek, E. L. Tarver, A. Lange, Chromatographia,24, 815 (1987).
A. G. MacDiarmid, Ed., Organometallic Compounds of the Group IV Elements. The Bond to Carbon, Marcel Dekker, New York, 1968, Vol. 1, Part I.
R. M. Smith, J. Chromatogr.,236, 313 (1982).
Rainin Dynamax Review,4, 5 (1990).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pesek, J.J., Rashet, W. Synthesis of a propyldimethylphenylsilane stationary phase using the allyl bonded phase as an intermediate. Chromatographia 30, 442–446 (1990). https://doi.org/10.1007/BF02328514
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02328514