Abstract
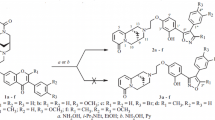
New 3-(2-benzothiazolyl)chromones were obtained by the reaction of 2-(2,4-dihydroxy-5-ethylphenacyl)-benzothiazole with triethyl orthoformate or carboxylic acid anhydrides. Aminomethylation of the chromones and also of familiar 3-(2-benzimidazolyl)chromones by substituted 1,1-diaminomethanes gave Mannich bases.
Similar content being viewed by others
References
M. S. Frasinyuk and V. P. Khilya, Khim. Geterotsikl. Soedin., No. 10, 1377 (1997).
P. Da Re and L. Verlicchi, Ann. Chim. Ital.,50, 1273 (1960).
P. Da Re, L. Verlicchi, and I. Setnikar, J. Org. Chem.,25, 1097 (1960).
N. V. Gorbulenko, M. S. Frasinyuk, Khim. Geterotsikl. Soedin., No. 4, 464 (1994).
M. S. Frasinyuk, N. V. Gorbulenko, and V. P. Khilya, Khim. Geterotsikl., Soedin., No. 9, 1237 (1997).
F. Kallay and G. Janzso, Tetrahedron Lett.,16, 1443 (1978).
F. Kallay, G. Janzso, and I. Egyed, Flavonoids, Bioflavonoids, Proc. 5th Hung. Bioflavonoid Symp. (1977), p. 235; Chem. Abs.,89, 43029 (1978).
Additional information
For Communication 21, see [1].
Taras Shevchenko Kiev University, Kiev, Ukraine. Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 8, pp. 1072–1078, August, 1998.
Rights and permissions
About this article
Cite this article
Frasinyuk, M.S., Turov, A.V. & Khilya, V.P. Chemistry of the hetero analogs of isoflavones. 22. Mannich reaction in the benzimidazole and benzothiazole analogs of isoflavones. Chem Heterocycl Compd 34, 923–928 (1998). https://doi.org/10.1007/BF02311328
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02311328