Abstract
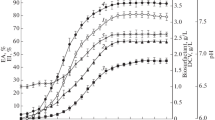
The nutritional requirements and growth characteristics of a biosurfactant-producingRhodococcus bacterium isolated from Kuwaiti soil were determined. Maximum cell yields (6.6 g/l) and biosurfactant production were achieved with a medium containing 2% (v/v)n-paraffin as a carbon and energy source, 0.2% lactose broth, optimal concentrations of nitrogen (nitrate), phosphorus, iron, magnesium and sodium sources, and minimal concentrations of potassium and trace element sources. The optimal pH was 6.8 for surfactant production and optimal temperature was 37°C. The biosurfactant produced after 16 to 33 h growth in a 7 I fermenter decreased both surface tension and interfacial tension of culture broth to below 27 and 1.8 mN/m, respectively, and was effective at critical micelle dilutions of 10−3. Data on biosurfactant biosynthesis suggest that the product is produced as a primary metabolite and, therefore, could be produced effectively under continuous fermentation conditions.
Similar content being viewed by others
References
Abu-Ruwaida, A.S., Banat, I.M. &Hamdan, I.Y. 1990 Chemostat optimization of biomass production of a mixed bacterial culture utilizing methanol.Applied Microbiology and Biotechnology 32, 550–555.
Akit, J., Cooper, D.G., Manninen, K.I. &Zajic, J.E. 1981 Investigation of potential biosurfactant production among phytopathogenicCorynebacterium and related soil microbes.Current Microbiology 6, 145–150.
Atlas, R.M. 1981 Microbial degradation of petroleum hydrocarbons: an environmental perspective.Microbiological Reviews 45, 180–209.
Brown, M.J., Foster, M., Moses, V., Robinson, J.P., Shales, S.W. &Springham, D.G. 1985 Novel biosurfactant for EOR. InProceedings of the Third European Meeting on Improved Oil Recovery pp. 241–248. Rome: AGIP S.P.A.
Chakrabarty, A.M. 1984 Genetically-manipulated microorganisms and their products in the oil service industries.Trends in Biotechnology 3, 32–38.
Cooper, D.G. &Goldenberg, B.G. 1987 Surface-active agents from twoBacillus species.Applied and Environmental Microbiology 53, 224–229.
Cooper, D.G. &Zajic, J.E. 1980 Surface compounds from microorganisms.Advances in Applied Microbiology 26, 229–256.
Cooper, D.G., MacDonald, C.R., Duff, J.B. &Kosaric, N. 1981a Enhanced production of surfactant fromBacillus subtilis by continuous product removal and metal cation addition.Applied and Environmental Microbiology 42, 408–412.
Cooper, D.G., Zajic, J.E. &Denis, C. 1981b Surface-active properties of a biosurfactant fromCorynebacterium lepus.Journal of the American Oil Chemists' Society 58, 77–80.
Cooper, D.G., Zajic, J.E. &Gerson, D.F. 1979 Production of surface-active lipids byCorynebacterium lepus.Applied and Environmental Microbiology 37, 4–10.
Desai, J.D. 1987 Microbial surfactants: evaluation, types, production and future application.Journal of Science and Industrial Research 46, 440–449.
Guerra-Santos, L., Kappeli, O. &Fiechter, A. 1984Pseudomonas aeruginosa biosurfactant production in continuous culture with glucose as carbon source.Applied and Environmental Microbiology 48, 301–305.
Guerra-Santos, L., Kappeli, O. &Fiechter, A. 1986 Dependence ofPseudomonas aeruginosa continuous culture biosurfactant production on nutritional and environmental factors.Applied Microbiology and Biotechnology 24, 443–448.
Haferburg, D., Hommel, R., Claus, R. &Kleber, H.P. 1986 Extracellular microbial lipids and biosurfactants. InAdvances in Biochemical Engineering/Biotechnology, Vol. 33, ed. Fiechter, A. pp. 53–93. Berlin: Springer.
Hisatsuka, K., Nakahara, T., Sano, N. &Yamada, K. 1971 Formation of rhamnolipid byPseudomonas aeruginosa and its function in hydrocarbon fermentation.Agricultural and Biological Chemistry 35, 686–692.
Ito, S. &Inoue, S. 1982 Sophorolipids fromTorulopsis bombicola possible relation to alkane uptake.Applied and Environmental Microbiology 43, 1278–1283.
Javaheri, M., Jenneman, G.E., McInerney, M.J. &Knapp, R.M. 1985 Anaerobic production of biosurfactant byBacillus licheniformis JF-2.Applied and Environmental Microbiology 50, 698–700.
Kaplan, N., Zosim, Z. &Rosenberg, E. 1987 Reconstitution of emulsifying activity ofAcinetobacter calcoaceticus BD4 emulsan by using pure polysaccharide and protein.Applied and Environmental Microbiology 53, 440–446.
Kappeli, O. &Finnerty, W.R. 1980 Characteristics of hexadecane partition by the growth medium ofAcinetobacter sp.Biotechnology and Bioengineering 22, 495–503.
Additional information
A.S. Abu-Ruwaida, S. Haditirto and A. Khamis are with the Kuwait Institute for Scientific Research, Biotechnology Department, P.O. Box 24885, 13109, Safat, Kuwait. I.M. Banat is now in Londonderry, Northern Ireland but was at the Kuwait Institute for Scientific Research at the time this paper was written. A.S. Abu-Ruwaida is the corresponding author.
In view of the annexation of Kuwait by Iraq in August 1990, this paper has been accepted without return to the author for attention to minor details. The Editor-in-Chief therefore assumes full responsibility for any errors or omissions.
Rights and permissions
About this article
Cite this article
Abu-Ruwaida, A.S., Banat, I.M., Haditirto, S. et al. Nutritional requirements and growth characteristics of a biosurfactant-producingRhodococcus bacterium. World J Microbiol Biotechnol 7, 53–60 (1991). https://doi.org/10.1007/BF02310920
Issue Date:
DOI: https://doi.org/10.1007/BF02310920