Abstract
Background: CA 19-9 levels are useful for the diagnosis of patients with pancreatic adenocarcinoma. However, interest has recently turned toward its use as a prognostic indicator. The purpose of this study is to determine whether postoperative CA 19-9 levels predict disease-free survival (DFS) and median survival (MS) in patients after resection.
Methods: Between 1988 and 1996, 40 patients underwent resection for pancreatic adenocarcinoma and were evaluated with postoperative CA 19-9 assays. Eight patients had low preoperative levels of CA 19-9 (<2) and were excluded.
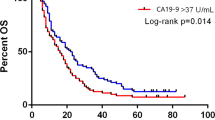
Results: CA 19-9 levels are good predictors of DFS and MS. Patients whose postoperative CA 19-9 values normalized by 3 to 6 months (<37 U/ml) had longer DFS (24 vs. 10 months,p<0.04) and MS (34 vs. 13 months,p<0.04). Patients with postoperative CA 19-9 values less than 180 U/ml at 1 to 3 months had a similar DFS (19 vs. 5 months,p<0.0009) and MS (34 vs. 13 months,p<0.0001) compared to patients with normal values at 3 to 6 months.
Conclusions: Postoperative measurements of CA 19-9 were the best predictors of DFS and MS. Values<180 U/ml at 3 months were as predictive as normal values by 3 to 6 months postoperatively. Consequently, CA 19-9 levels should be obtained for use as a stratification parameter in phase III trials.
Similar content being viewed by others
References
Parker SL, Tong T, Bolden S and Wingo PA. Cancer statistics.CA C J Clin 1996;46:5–27.
Gudjonsson B. Cancer of the pancreas. 50 years of surgery.Cancer 1987;60:2284–2303.
Trede M, Schwall G, Saeger HD. Survival after pancreaticoduodenectomy. 118 consecutive resections without an operative mortality.Ann Surg 1990;211:447–58.
Cameron JL, Crist DW, Sitzmann JV, Hruban RH, Boitnott JK, Seidler AJ, Coleman J. Factors influencing survival after pancreaticoduodenectomy for pancreatic cancer.Am J Surg 1991;161:120–5.
Steinberg W. The clinical utility of the CA 19-9 tumor-associated antigen.Am J Gastroenterol 1990;85:350–5.
Magnani JL, Steplewski Z, Koprowski H, Ginsberg V. Identification of the gastrointestinal and pancreatic cancer-associated antigen detected by monoclonal antibody 19-9 in the sera of patients as a mucin.Cancer Res 1983;43:5489–92.
Tempero MA, Uchida E, Takasaki H, Burnett DA, Steplewski Z, Pour PM. Relationship of carbohydrate antigen 19-9 and Lewis antigens in pancreatic cancer.Cancer Res 1987;47:5501–3.
Koprowski H, Steplewski Z, Mitchell K, Herlyn M, Herlyn D, Fuhrer P. Colorectal carcinoma antigens detected by hybridoma antibodies.Somat Cell Mol Genet 1979;5:957–71.
Del Villano BC, Brennan S, Brock P, et al. Radioimmunometric assay for a monoclonal antibody-defined tumor marker, CA 19-9.Clin Chem 1983;29:549–52.
Haglunde C, Roberts PJ, Kuusela P, Scheinin TM, Makela O, Jalanko H. Evaluation of CA 19-9 as a serum tumor marker in pancreatic cancer.Br J Cancer 1986;53:197–202.
Satake K, Chung YS, Umeyama K, Takeuchi T, Kim YS. The possibility of diagnosing small pancreatic cancer (less than 4.0 cm) by measuring various serum tumor markers.Cancer 1991;68:149–52.
Glenn J, Steinberg WM, Kurtzman SH, Steinberg S, Sindelar WF. Evaluation of the utility of a radioimmunoassay for serum CA 19-9 levels in patients before and after treatment of carcinoma of the Pancreas.J Clin Oncol 1988;6:462–8.
Sperti C, Pasquali C, Catalini S, Cappellazzo F, Bonadimani B, Behboo R, Pedrazzoli S. CA 19-9 as a prognostic index after resection for pancreatic cancer.J Surg Oncol 1993;52:137–41.
Beretta E, Malesci A, Zerbi A, et al. Serum CA 19-9 in the post-surgical follow-up of patients with pancreatic cancer.Cancer 1987;60:2428–31.
Ishikawa O, Ohigashi H, Imaoka S, et al. Preoperative indications for extended pancreatectomy for locally advanced pancreas cancer involving the portal vein.Ann Surg 1992;215:231–6.
American Joint Committee on Cancer. Exocrine pancreas. In: Manual for Staging of Cancer, 4th ed. Philadelphia: JB Lippincott, 1992:109–11.
Hoffman JP, Weese JL, Solin LJ, et al. A pilot study of preoperative chemoradiation for patients with localized adenocarcinoma of the pancreas.Am J Surg 1995;169:71–8.
Hoffman JP, Weese JL, Lipsitz S, Solin LJ, Benson AB III, Pisansky TM, Dalton A. Preoperative chemoradiation for patients with resectable pancreatic adenocarcinoma. An Eastern Cooperative Oncology Group (ECOG) phase II study.Proc Am Soc Clin Oncol 1995;14:201. Abstract.
Winek T, Hamre D, Mozell E, Vetto M. Prognostic factors for survival after pancreaticoduodenectomy for malignant disease.Am J Surg 1990;159:454–6.
Moossa AR, Levin B. The diagnosis of “early” pancreatic cancer: The University of Chicago experience.Cancer 1981;47:1688–97.
Pearce S, Thornes H, Carr D, Tanner A. Diagnostic pitfall: interpretation of CA 19-9 concentrations in the presence of hepatic dysfunction.Gut 1994;35:707–8.
Willett CG, Daley WJ, Warshaw AL. CA 19-9 is an index of response to neoadjunctive chemoradiation therapy in pancreatic cancer.Am J Surg 1996;172:350–2.
Lundin J, Roberts PJ, Kuusela P, Haglund C. Prognostic significance of serum CA 242 in pancreatic cancer. A comparison with CA 19-9.Anticancer Res 1995;15:2181–6.
van den Bosch RP, van Eijck CHJ, Mulder PGH, Jeekel J. Serum CA 19-9 determination in the management of pancreatic cancer.Hepatogastroenterology 1996;43:710–3.
Pohl AL. Surveillance of cancer patients with tumor markers.J Tumor Marker Oncol 1987;2(1):1–12.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Montgomery, R.C., Hoffman, J.P., Riley, L.B. et al. Prediction of recurrence and survival by post-resection CA 19-9 values in patients with adenocarcinoma of the pancreas. Annals of Surgical Oncology 4, 551–556 (1997). https://doi.org/10.1007/BF02305535
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02305535