Abstract
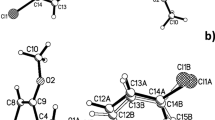
Bromination of 2,4-substituted 1,2-dihydro-4H-benzoxazines with bromine in acetic acid was conducted. It was shown that either the corresponding 6,8-dibromo-1,2-hydrobenzoxazines or the products of their dehydrogenation — 6,8-dibromobenzoxazines, are primarily formed as a function of the structure of the dihydrobenzoxazine and the concentration of bromine in the reaction mixture. The structure of 6,8-dibromo-2-(5-nitrofuryl-2)-4, 4-diphenyl-1,2-dihydro-4H-3,1-benzoxazine was investigated by XSA. A stacking interaction between the nitrofuran fragment of one molecule and the condensed benzene ring of the other was detected in the crystal.
Similar content being viewed by others
References
E. V. Gromachevskaya, T. P. Kosulina, F. V. Kvitkovskii, and V. G. Kul'nevich, Khim. Geterotsikl. Soedin., No. 6, 841 (1997).
E. V. Gromachevskaya, V. O. Loginova, and A. A. Kovaleva, Chemistry and Technology of Furan Compounds [in Russian], Mezhvuz. Sb. Nauch. Tr. Krasnodar Politekhn. In-ta, Krasnodar (1987), p. 45.
GB Patent No. 2,053,900; A. Berge, G. Niels, P. Herbert, Chem. Abstr.,94, 121077 (1981).
N. S. Zefirov and V. A. Palyulin, Dokl. Akad. Nauk,252, 111 (1980).
G. M. Sheldrick, Computational Crystallography, Oxford University Press, New York-Oxford (1982), p. 506.
E. V. Gromachevskaya, V. G. Kul'nevich, T. P. Kosulina, and V. S. Pustovarov, Khim. Geterotsikl. Soedin., No. 6, 842 (1988).
Additional information
See [1] for Communication 12.
Kuban State Technological University, Krasnodar 350072. Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1391–1397, October, 1997.
Rights and permissions
About this article
Cite this article
Gromachevskaya, E.V., Krapivin, G.D., Zavodnik, V.E. et al. Studies of 4H-3,1-benzoxazines. Chem Heterocycl Compd 33, 1209–1214 (1997). https://doi.org/10.1007/BF02290872
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02290872