Abstract
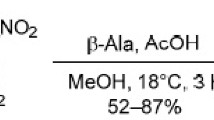
The synthesis of α-bromo-4-difluoromethoxy- and α-bromo-2,4-bis(difluoromethoxy)acetophenones has been carried out. Conditions have been studied for condensing the latter with cyclic tri- and tetramethylenamidinines and with pyridine-containing derivatives. A series of pyrrolo[1,2-a]imidazole and imidazo[2,1-a]pyridine derivatives containing difluoromethoxy groups has been synthesized.
Similar content being viewed by others
References
P. E. Bender, US Patent 4,186,205; Chem. Abs.,92, 181195 (1980).
A. Andreani, M. Rambaldi, and G. Locatelli, Eur. J. Med. Chem.,29 339 (1994).
Y. Morisawa, T. Okada, T. Okazoe, N. Nakamura, Y. Inoe, and H. Ebisu, Japanese Patent 94,184,148; Chem. Abs.,121, 300893 (1994).
G. B. Barlin, L. P. Davies, and P. W. Harrison, Austral. J. Chem.,48, 1031 (1995).
L. M. Yagupol'skii, Aromatic and Heterocyclic Compounds with Fluorine-Containing Substituents [in Russian], Naukova Dumka, Kiev (1988), p. 319.
V. V. Kastron, R. O. Vitolin', G. Ya. Dubur, I. P. Skrastin'sh, and A. A. Kimenis, Khim.-farm. Zh.,21, 554 (1987).
V. V. Kastron, R. O. Vitolin', Yu. A. Fialkov, S. V. Shelyazhenko, G. Ya. Dubur, A. A. Kimenis, and L. M. Yagupol'skii, Authors Certificate 706,410 USSR; Byull. Izobret., No. 48, 88 (1979).
Yu. A. Fialkov and S. V. Shelyazhenko, Authors Certificate 1,085,971 USSR; Byull. Izobret., No. 14, 79 (1984).
S. V. Shelyazhenko, Yu. A. Fialkov, and L. M. Yagupol'skii, Zh. Org. Khim.,28, 1652 (1992)
E. J. Moriconi and A. A. Cevasco, J. Org. Chem.,33, 2109 (1968).
Additional information
T. G. Shevchenko Chernigov Pedagogical Institute, Chernigov 250038. Institute of Organic Chemistry, National Academy of Sciences of Ukraine, Kiev 252660. Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1371–1376, October, 1997.
Rights and permissions
About this article
Cite this article
Demchenko, A.M., Nazarenko, K.G., Fedyuk, D.V. et al. Preparation and use ofα-bromomono- and-bromobis-difluoromethoxyacetophenones in the synthesis of polymethyleneimidazoles with an angular nitrogen atom. Chem Heterocycl Compd 33, 1191–1195 (1997). https://doi.org/10.1007/BF02290868
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02290868