Summary
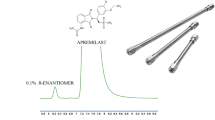
The native enantioselectivity in binding of human serum albumin (HSA) towards 2-aryl propionic acid non-steroidal anti-inflammatory drugs (2-APA-NSAIDs, the “profens”) was found to be preserved when the protein was immobilized within a commercially available diol high-performance liquid chromatographic column. High capacity factors were obtained, reflecting the previously observed extensive binding of the 2-APA-NSAIDs to free HSA. The capacity factors were modified by the addition of octanoic acid to the mobile phase. Chiral resolution of the enantiomers of all nine 2-APA-NSAIDs studied was achieved. Preliminary studies show that in addition to being a useful chiral analytical tool for this therapeutically important series of compounds, the HSA chiral stationary phase may provide useful information on the affinity and binding mechanism of small molecules to HSA.
Similar content being viewed by others

References
R. D. Knihinicki, K. M. Williams, R. O. Day, Bioch. Pharmacol.,38, 4389 (1989).
F. Jamali, B. W. Berry, M. R. Tehrani, A. S. Russel, J. Pharm. Sci.,77, 666 (1988).
J. Gal, in “Drug Stereochemistry: Analytical Methods and Pharmacology”,I. W. Wainer andD. E. Drayer, eds., Marcel Dekker, New York, 1988, p. 77.
I. W. Wainer, T. D. Doyle, J. Chromatogr.,284, 117 (1984).
Y. Okamoto, R. Aburatani, Y. Kaida, K. Hatada, N. Inotsume, M. Nakano, Chirality,1, 239 (1989).
S. Allenmark, S. Andersson, Chirality,1, 154 (1989).
J. Hermansson, M. Eriksson, J. Liq. Chromatogr.,9, 621 (1986).
T. Miwa, T. Miyakawa, M. Kayano, Y. Miyake, J. Chromatogr.,408, 315–322 (1987).
E. Domenici, C. Bertucci, P. Salvadori, G. Felix, I. Cahagne, S. Motellier, I. W. Wainer, Chromatographia,29, 170 (1990).
E. Domenici, C. Bertucci, P. Salvadori, I. W. Wainer, J. Pharm. Sci., in press.
E. Domenici, C. Bertucci, P. Salvadori, S. Motellier, I. W. Wainer, Chirality, in press.
J. H. Lin, D. M. Cochetto, D. E. Duggan, Clin. Pharmacokinet.,12, 402 (1987).
A. M. Evans, R. L. Nation, L. N. Sansom, F. Bochner, A. A. Somogyi, Eur. J. Clin. Pharmacol.,36, 283 (1989).
S. Wanwimolruk, D. J. Birkett, P. M. Brookes, Mol. Pharmacol.,24, 458 (1983).
I. Sjöholm, B. Ekman, A. Kober, I. Ljungstedt-Påhlman, B. Seiving, T. Sjödin, Mol. Pharmacol.,16, 767 (1979).
I. Sjöhlm, in “Drug-Protein Binding”,M. M. Reidenberg andS. Erill, eds., Praeger, New York, 1986, p. 36.
E. J. D. Lee, K. Williams, R. Day, G. Graham, D. Champion, Br. J. Clin. Pharmacol.,19, 669 (1985).
K. J. Fehske, W. E. Müller, U. Wollert, Bioch. Pharmacol.,30, 687 (1981).
W. E. Müller, in “Drug Stereochemistry: Analytical Methods and Pharmacology”,I. W. Wainer andD. E. Drayer, eds., Marcel Dekker, New York, 1988, p. 227.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Noctor, T.A.G., Felix, G. & Wainer, I.W. Stereochemical resolution of enantiomeric 2-aryl propionic acid non-steroidal anti-inflammatory drugs on a human serum albumin based high-performance liquid chromatographic chiral stationary phase. Chromatographia 31, 55–59 (1991). https://doi.org/10.1007/BF02290496
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02290496