Abstract
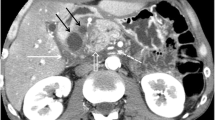
To verify the influence of obstructive jaundice on pancreatic growth, the anteroposterior width of the pancreas was measured by computed tomography in 30 cholangiocarcinoma patients excluded patients with distal bile duct tumor (jaundice group) and 74 control subjects. Follow-up examinations were performed on 12 patients with and without internal biliary drainage to elucidate the temporal relationship between pancreatic enlargement and the diversion of the obstructed biliary stream. Histologic analysis on autopsy samples from 13 control and 10 jaundice cases also was performed. Mean pancreatic head and body widths in the jaundice group were 2.93±0.3 cm and 2.01±0.3 cm, respectively. These values were significantly greater than those of the controls (2.13±0.3 cm and 1.49±0.3 cm,P<0.01). The glandular widths returned to their normal sizes following internal biliary drainage. No changes were seen in patients who underwent external drainage alone. Histologic examination revealed that enlargement of the acinar cells or of the islet of Langerhans was often seen in the jaundiced patients. Therefore obstructive jaundice is thought to cause pancreatic growth through a trophic effect by interrupting biliary circulation.
Similar content being viewed by others
References
Chernick SS, Lepkovsky S, Chaikoff LL: A dietary factor regulating the enzyme content of the pancreas; changes induced in size and proteolytic activity of the chick pancreas by the ingestion of raw soybean meal. Am J Physiol 155:33–41, 1948
Baba N, Suzuki T, Tobe T, Inoue K, Chowdhury P, Chang LW, Rayford P: Influence of obstructive jaundice on pancreatic growth and on basal plasma levels of cholecystokinin and gastrin in rats. Dig Dis Sci 31:1233–1241, 1986
Green GM, Nasset ES: Effect of bile duct obstruction on pancreatic enzyme secretion and intestinal proteolytic enzyme activity in the rats. Dig Dis 22:437–444, 1977
Newman BM, Lee PC, Brooks S, Cooney DR, Lebenthal E: Enteral feedback control of pancreatic hypertrophy: The role of pancreatic biliary secretions. Proc Soc Exp Biol Med 181:226–232, 1986
Brand SJ, Morgan RHG: Stimulation of pancreatic secretion and growth in the rat after feeding cholestyramine. Gastroenterology 83:851–859, 1982
Muranaka T: Morphologic changes in the body of the pancreas secondary to a mass in the pancreatic head. Analysis by CT. Acta Radiol 31:483–488, 1990
Muranaka T, Kato A, Matsuura Y, Hanada K, Nakano I, Yoshida T, Toda T, Oshiumi Y: Enlargement of the pancreas in non-alcoholic cirrhosis: A computed tomography and pathological investigation. Eur Radiol 2:99–104, 1992
Kolmanskog F, Swenson T, Vatin MH, Larsen S: Computed tomography and ultrasound of the normal pancreas. Acta Radiol Diagn 23:443–451, 1982
Ferrucci JT Jr, Wittenberg J, Black EB, Kirkpatrik RH, Hall DA: Computed body tomography in chronic pancreatitis. Radiology 130:175–182, 1976
Neff CC, Simeone JF, Wittenberg J, Mueller PR, Ferrucci JT, Jr: Inflammatory pancreatic masses: problems in differentiating focal pancreatitis from carcinoma. Radiology 150:35–38, 1984
Silverstein W, Isikoff MB, Hill MC, Barkin J: Diagnostic imaging of acute pancreatitis: Prospective study using CT and sonography: AJR 137:497–502, 1981
Muranaka T, Teshima K, Honda H, Nanjo T, Hanada K, Oshiumi Y: Computed tomography and histologic appearance of pancreatic metastases from distant sources. Acta Radiol 30:615–619, 1989
Booth AN, Robbins DJ, Ribelin WE, Deeds F: Effect of raw soybean meal and amino acids on pancreatic hypertrophy in rats. Proc Soc Exp Biol Med 104:681–683, 1960
Booth AN, Robbins DJ, Ribelin WE, Deeds F, Smith AK, Rackis JJ: Prolonged hypertrophy and reversibility in rats fed raw soybean meal. Proc Soc Exp Biol Med 116:1067–1069, 1964
Yanatori Y, Fujita T: Hypertrophy and hyperplasia in the endocrine and exocrine pancreas of rats fed soybean trypsin inhibitor or repeatedly injected with pancreozymin. Arch Histol Jpn 39:67–78, 1976
Sirakawa M: Biochemical studies on the time limit for surgical release of obstructive jaundice after common bile duct ligation in rats and dogs. Jpn Surg Gastroenterol 11:359–368, 1978
Schaffuer R, Bacchia PG: Mechanism of cholestasis. Gastroenterology 60:888–897, 1971
Langier R, Sarles H: Action of olcic acid on the exocrine pancreatic secretion of the conscious rat: evidence for anticholecystokinin pancreozymin factor. J Physiol (London) 271:81–92, 1977
Singer MV, Solomon TE, Wood J, Grossman MI: Latency of pancreatic enzyme response to intraduodenal stimulus. Am J Physiol 238:23–29, 1980
Green GM, Lyman RL: Feedback regulation of pancreatic enzyme secretion as a mechanism for tripsin inhibitor-induced hypersecretion in rats. Proc Soc Exp Biol Med 140:6–12, 1972
Miyasaka K, Kurosawa H, Kitani K: Proglumide stimulate basal pancreatic secretion in the conscious rat. Digestion 37:135–143, 1987
Folsch UR, Winckler K, Wormsley KG: Influence of repeated administration of cholecystokinin and secretin of the pancreas of the rat. Scand J Gastroenterol 13:663–671, 1978
Dowling RH, Hosomi M, Stace NH, Lirussi F, Miazza B, Levan H, Murphy GM: Hormones and polyamines in intestinal and pancreatic adaptation. Scand J Gastroenterol 20:84–95, 1985
Morisset J, Benrezza KD: Polyamines and pancreatic growth induced by caerulein. Life Sci 35:2471–2480, 1984
Johnson LR: Effects of gastrointestinal hormones on pancreatic growth. Cancer 47:1640–1645, 1981
Mainz DL, Black O, Webster PD: Hormone control of pancreatic growth. J Clin Invest 52:2300–2304, 1973
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Muranaka, T., Ro, T., Masuda, Y. et al. Pancreatic enlargement in obstructive jaundice. Digest Dis Sci 40, 1554–1560 (1995). https://doi.org/10.1007/BF02285208
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02285208