Abstract
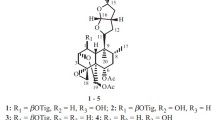
Two new clerodanolides — salvicinolide and salvicinolin — have been isolated from the epigeal part ofPulicaria salviifolia Bge inMem (fam. Compositae). Their structures have been established by an investigation of their spectral and physicochemical characteristics. It has been found that salvicinolide is capable of cyclizing at the C-6 and C-19 carbon atoms with the formation of a second lactone ring. The structure of 6α-hydroxy-19-carboxy-trans-clero-3,13-dien-15-olide is proposed for salvicinolide, and 6α,13-dihydroxy-19-carboxy-rans-cleroda-3,13-diene for salvicinolin.
Similar content being viewed by others
References
G. V. Sagitdinova, K. A. Éshbakova, and V. M. Malikov, Khim. Prir. Soedin., 246 (1994).
M. R. Nurmukhamedova, Sh. Z. Kasimov, N. D. Abdullaev, and G. P. Yagudaev, Khim. Prir. Soedin., 201 (1985).
M. R. Nurmukhamedova, N. D. Abdullaev, and G. P. Sidyakin, Khim. Prir. Soedin., 299 (1986).
B. Tashkhodzhaev, I. M. Yusupova, A. I. Yankovskaya, M. R. Nurmukhamedova, Yu. T. Struchkov, and M. R. Yagudaev, Khim. Prir. Soedin., 63 (1988).
Additional information
Institute of the Chemistry of Plant Substances, Academy of Sciences of the Republic of Uzbekistan, Tashkent fax (3712) 40 64 75. Translated from Khimiya Prirodnykh Soedinenii, No. 4, pp. 594–598, July–August, 1997.
Rights and permissions
About this article
Cite this article
Éshbakova, K.A., Sagitdinova, G.V., Levkovich, M.G. et al. Diterpenoids ofPulicaria salviifolia IV. Structures of salvicinolide and salvicinolin. Chem Nat Compd 33, 458–461 (1997). https://doi.org/10.1007/BF02282365
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02282365