Summary
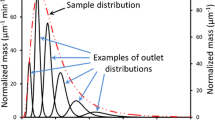
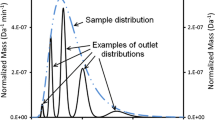
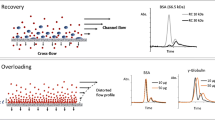
Several types of membrane have been tested for use in organic solvent flow field-flow fractionation in an asymmetric channel. The practical problems most commonly encountered were leakage of air and solvent through the support layer on which the membranes are cast, and unequal swelling of the membrane and the support layer in the organic solvent, leading to ridging of the membrane in the channel. Three types of membrane were found suitable for the separation of polystyrene standards with tetrahydrofuran as solvent. The best results were obtained with a fluoropolymer membrane. Fair agreement was found between theory and practice for the dependence of retention times on the relative molecular mass of the standards and on the flow regime. Use of scanning electron microscopy revealed that for a number of the membrane materials some pores were much larger than expected on the basis of the indicated molecular weight cut-off. Whereas these materials could not be used for the fractionation of soluble polymers, they could be applied with some success to the separation of solid latex and silica particles. A PTFE membrane could be used for the separation of latexes and silica particles suspended in acetonitrile as carrier liquid. In general, however, the retention times of these particles were shorter than theoretically predicted.
Similar content being viewed by others
References
J. C. Giddings, Science260, 1456 (1993).
A. C. van Asten, W. Th. Kok, R. Tijssen, H. Poppe, J. Polym. Sci., Part B,34, 297 (1996).
G. Liu, J. C. Giddings, Chromatographia34, 483 (1992).
S. L. Brimhall, M. N. Myers, K. D. Caldwell, J. C. Giddings, Polym. Mater. Sci. Eng.50, 48 (1984).
K. D. Caldwell, S. L. Brinhall, Y. Gao, J. C. Giddings, J. Appl. Polym. Sci.36, 703 (1988).
J. J. Kirkland, C. H. Dilks Jr., Anal. Chem.64, 2836 (1992).
M. E. Miller, H. Lee, X. Li, R. Szentirmay, J. C. Giddings, Polym. Prepr.35, 764 (1994).
H. L. Lee, J. F. G. Reis, J. Dohner, E. N. Lightfoot, AIChE J.20, 776 (1974).
J. Å. Jönsson, A. Carlshaf, Anal. Chem.61, 11 (1989).
J. E. G. J. Wijnhoven, J. P. Koorn, H. Poppe, W. Th. Kok, J. Chromatogr. A699, 119 (1994).
J. E. G. J. Wijnhoven, J. P. Koorn, H. Poppe, W. Th. Kok, J. Chromatogr. A, in press.
R. Tijssen, J. Bos, M. E. van Kreveld, Anal. Chem.58, 3036 (1986).
J. C. Giddings, P. S. Williams, M. A. Benincasa, J. Chromatogr.626, 23 (1992).
K.-G. Wahlund, J. C. Giddings, Anal. Chem.59, 1332 (1987).
W. Mandema, H. Zeldenrus, Polymer18, 835 (1977).
C. Tanford, “Physical Chemistry of Macromolecules”, John Wiley and Sons, New York, 1961, Chapter 6.
M. N. Meyers, J. C. Giddings, Anal. Chem.54, 2284 (1982).
S. K. Ratanathanawongs, J. C. Giddings, ACS Symp. Series521, 13 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wijnhoven, J.E.G.J., van Bommel, M.R., Poppe, H. et al. Practical experience with organic solvent flow field-flow fractionation. Chromatographia 42, 409–415 (1996). https://doi.org/10.1007/BF02272132
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02272132