Summary
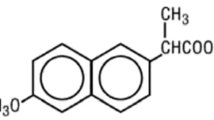
An HPLC method has been developed for the determination of SL 85.1016, a new calcium antagonist arylbenzylamide methylthioether derivative. SL 85.1016 and the internal standard, SL 87.0210, are extracted from alkaline human plasma withn-hexane and back extracted into 0.05 M phosphate buffer (pH 2.5; 0.2 ml). Acetonitrile (50 μl) is added to the final aqueous extract in order to prevent absorption of the compounds of interest onto the walls of the glass tube; this solution then is partially processed by HPLC on a C18 column with UV detection (254 nm). The determination limit of the method is 2 ng.ml−1 of SL 85.1016 in human plasma; the response to the drug is linear in the range 2–200 ng.mg−1.
Similar content being viewed by others
References
D. M. Watterson, F. F. Vincenzi, Eds. Ann. N. Y. Acad. Sci.356, 455 (1980).
Y. M. Lin, Mol. Cell Biochem.45, 101 (1982).
M. Chaffman, R. N. Broyden, Drugs29, 387 (1985).
S. C. Montamat, D. R. Abernethy, Clin. Pharmacol. Ther.45, 682 (1989).
R. A. Boyd, S. K. Chin, O. Don Pedro, D. Verotta, L. B. Sheiner, R. L. Williams, K. M. Giacomini, Eds., Clin. Pharmacol. Ther.46, 408 (1989).
J. G. Kelly, K. O'Malley, Clin. Pharmacokinet.22 (6), 416 (1992).
J. Duchier, A. Cournot, G. Bianchetti, C. Dubruc, L. Darchy, P. Rosenzweig, Recherche (L.E.R.S.), Paris, France: Internal Report M 35.03.89.21 (1990).
J. A. Rouchouse, M. Manoha, J. T. Burke, Synthélabo Recherche (L.E.R.S.), Paris, France: Internal Report M 38.02.88.005 (1988).
P. Hartvig, J. Vessman, Anal. Lett.7, 223 (1974).
P. Hartvig, N. O. Ahnfelt, K. E. Karlsson, Acta Pharm. Suecica13, 181 (1976).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Flaminio, L., Malavasi, B., Ripamonti, M. et al. Determination of a new calcium antagonist in human plasma by liquid extraction enrichment and HPLC with UV detection. Chromatographia 36, 343–346 (1993). https://doi.org/10.1007/BF02263888
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02263888