Abstract
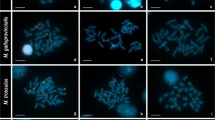
In the present investigation chromosomal preparations ofAsellus aquaticus were sequentially stained with chromomycin A3 to reveal the heterochromatic areas, hybridizedin situ with rDNA probes in order to map the ribosomal genes and finally silver stained to check the transcriptional activity of these genes. The results indicate the existence of a substantial correspondence of location and size among the heterochromatic regions and the regions over which thein situ hybridization signals spread. The ribosomal genes, quite independently of their location in the secondary constriction, can be silver stained and thus appear to be transcriptionally active. The ribosomal sequences also hybridize to the entire heterochromatic areas observed on the probable Y chromosome identified in some males of a natural population. These rRNA genes are only rarely transcriptionally active.
Similar content being viewed by others
References
Di Castro M, Prantera G, Cipriani L, Rocchi A (1983) Silver staining analysis of nucleolar-organizer activity during spermatogenesis ofAsellus aquaticus (Crustacea Isopoda).Genetica 60: 163–166.
Di Castro M, Prantera G, Marchetti E, Rocchi A (1977) N-banding and nucleolus organizers inAsellus aquaticus (Crust. Isop.).Heredity 39: 259–262.
Fitch DHA, Strausbaugh LD, Barret V (1990) On the origins of tandemly repeated genes: Does histone gene copy number inDrosophila reflect chromosomal location?.Chromosoma 99: 118–124.
Howell WM, Black DA (1980) Controlled silver-staining of nucleolus organizer regions with a protective colloidal developer: a 1-step method.Experientia 36: 1014.
Jordan G (1987) At the heart of the nucleolus.Nature 329: 489–490.
Medina FJ, Risueño MC, Sanchez-Pina MA, Fernández-Gómez ME (1983) A study on nucleolar silver staining in plant cells. The role of argyrophilic proteins in nucleolar physiology.Chromosoma 88: 149–155.
Miller DA, Dev VG, Tantravahi R, Miller OJ (1976) Suppression of human nucleolus organizer activity in mouse-human somatic hybrid cells.Exp Cell Res 101: 235–243.
Pardue ML, Brown DD, Birnstiel ML (1973) Location of the genes for 5S ribosomal RNA inXenopus laevis.Chromosoma 42: 191–203.
Pendás AM, Morán P, Garcia-Vázquez E (1993a) Ribosomal RNA genes are interspersed throughout a heterochromatic chromosome arm in Atlantic salmon.Cytogenet Cell Genet 63: 128–130.
Pendás AM, Morán P, Garcia-Vázquez E (1993b) Multi-chromosomal location of ribosomal RNA genes and heterochromatin association in brown trout.Chrom Res 1: 63–67.
Rocchi A, Prantera G, Di Castro M (1980) A study of the heterochromatin ofAsellus aquaticus (Crust. Isop.)Caryologia 33: 401–409.
Rocchi A, Prantera G, Lanza V, Di Castro M (1984) Incipient sex chromosome differentiation in an isopod crustacean species,Asellus aquaticus.Chromosoma 89: 193–196.
Salvadori S, Diana AM, Colucci E, Floridia G, Rossi E, Zuffardi O (1995) Colocalization of (TTAGGG)n telomeric sequences and ribosomal genes in Atlantic eels.Chrom Res 3: 54–58.
Schmid M (1982) Chromosome banding in Amphibia. VII Analysis of the structure and variability of NORs in Anura.Chromosoma 87: 327–344.
Stephenson EC, Erba HP, Gall JG (1981) Histone gene clusters of the newtNotophthalmus are separated by long tracts of satellite DNA.Cell 24: 639–647.
Volpi EV, Pelliccia F, Lanza V, Di Castro M, Rocchi A (1992) Morphological differentiation of a sex chromosome and ribosomal genes inAsellus aquaticus (Crust. Isop.).Heredity 69: 478–482.
Author information
Authors and Affiliations
Additional information
accepted for publication by M. Schmid
Rights and permissions
About this article
Cite this article
Barzotti, R., Pelliccia, F. & Rocchi, A. Heterochromatin and ribosomal genes inAsellus aquaticus (Crust. Isop.). Chromosome Res 4, 177–181 (1996). https://doi.org/10.1007/BF02254955
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02254955