Abstract
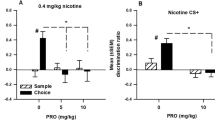
We have investigated the effects of environmental conditioning on the induction of nicotine sensitization of locomotion, stereotypy and nucleus accumbens dopamine release. Sprague-Dawley rats, some of which had been previously implanted with a microdialysis guide cannula over the nucleus accumbens, were sensitized with 5 days of repeated nicotine (0.6 mg/kg per day, SC) or saline injections (1 ml/kg per day). During nicotine treatment the drug administration was either paired with the microdialysis/activity monitor testing chamber (conditioned) (n=6) or with the animal's home cage (unconditioned) (n=6) and after 60 min the animal was returned to home cage and received a second injection of saline 15 min later. A third group received saline in the testing apparatus followed by nicotine in the home cage (pseudo-conditioned) (n=6). In the guide cannulated animals, 2 mm microdialysis probes were inserted after completing day 5 of treatment and all animals were tested for their response to nicotine (0.6 mg/kg, SC) on day 6. Both locomotor activity and nucleus accumbens dopamine release showed a larger response subsequent to nicotine challenge in the nicotine versus saline pretreated animals in the conditioned group, but not in the unconditioned group. In the pseudo-conditioned group there was an increase in the stereotypy responses to nicotine, however the locomotor and dopamine release responses were not significantly enhanced. The results from the conditioned group were confirmed in animals which were tested for behavioral activation and dopamine release simultaneously (n=5). These findings indicate that nicotine sensitization of locomotor activity and nucleus accumbens dopamine release (using a 5-day pretreatment protocol) is dependent on conditioning the animal to the testing environment during nicotine pretreatment.
Similar content being viewed by others
References
Balfour DJK, Benwell MEM, Vale AL (1991) Studies on the role of mesolimbic dopamine in behavioral responses to chronic nicotine. In: Adlkofer F, Thurau K (eds) Effects of nicotine on biological systems. Birkhäuser, Berlin, pp 407–416
Benwell MEM, Balfour DJK (1992) The effects of acute and repeated nicotine treatment on nucleus accumbens dopamine and locomotor activity. Br J Pharmacol 105: 849–856
Brown EE, Robertson GS, Fibiger HC (1992) Evidence for conditioned neuronal activation following exposure to a cocainepaired environment: role of forebrain limbic structures. J Neurosci 12: 4112–4121
Clarke PBS (1990) Commentary — dopaminergic mechanisms in the locomotor stimulant effects of nicotine. Biochem Pharmacol 40: 1427–1432
Clarke PBS, Kumar R (1983a) Characterization of the locomotor stimulant action of nicotine in tolerant rats. Br J Pharmacol 80: 587–594
Clarke PBS, Kumar R (1983b) The effects of nicotine on locomotor activity in non-tolerant and tolerant rats. Br J Pharmacol 78: 329–337
Clarke PBS, Pert A (1985) Autoadiographic evidence for nicotine receptors on nigrostriatal and mesolimbic dopaminergic neurons. Brain Res 348: 355–358
Clarke PBS, Fu DS, Jakubovic A, Fibiger HC (1988) Evidence that mesolimbic dopaminergic activation underlies the locomotor stimulant action of nicotine in rats. J Pharmacol Exp Ther 246: 701–708
Cline EJ, Ksir C (1988) Nicotine effect on locomotor activity is not reduced by dopamine receptor blockade. Soc Neurosci Abstr 14: 1137
Corrigall WA, Coen KA (1991) Selective dopamine antagonists reduce nicotine self-administration. Psychopharmacology 104: 171–176
Costall B, Naylor RJ (1977) Mesolimbic and extrapyramidal sites for the mediation of stereotyped behavior patterns and hyperactivity by amphetamine and apomorphine in the rat. In: Ellinwood EH, Kilbey MM (eds) Cocaine and other stimulants. Plenum Press, New York, pp 47–76
Crawford CA, McDougal SA, Bolanos CA, Hall S, Berger SP (1995) The effects the kappa agonist U-50,488 on cocaine-induced conditioned and unconditioned behaviors and Fos immunoreactivity. Psychopharmacology 120: 392–399
Damsma G, Day J, Fibiger HC (1989) Lack of tolerance to nicotine-induced dopamine release in the nucleus accumbens. Eur J Pharmacol 168: 363–368
Dietze S, Kuschinsky K (1994) Effect of conditioning withd-amphetamine on the extracellular concentration of dopamine and metabolites in the striatum of behaving rats. Naunyn-Schmiedeberg's Arch Pharmacol 350: 22–27
Fontana DJ, Post RM, Pert A (1993) Conditioned increases in mesolimbic dopamine overflow by stimuli associated with cocaine. Brain Res 629: 31–39
Fung YK (1989) Effects of chronic nicotine pretreatment on (+)-amphetamine and nicotine-induced synthesis and release of [3H] dopamine from [3H] tyrosine in rat nucleus accumbens. J Pharm Pharmacol 41: 66–68
Fung YK, Lau Y-S (1989) Effect of nicotine pretreatment on striatal dopaminergic system in rats. Pharmacol Biochem Behav 32: 221–226
Goldberg SR, Spealman RD, Goldberg DM (1981) Persistent high rate behavior maintained by intravenous self administration of nicotine. Science 214:573–575
Grenhoff J, Aston-Jones G, Svensson T (1986) Nicotinic effects on the firing pattern of midbrain dopamine neurons. Acta Physiol Scand 128:351–358
Henningfield JE, Goldberg SR (1983) Control of behavior by intravenous nicotine injections in human subjects. Pharmacol Biochem Behav 19:1021–1026
Hernandez L, Hoebel BG (1990) Feeding enhances dopamine turnover in the prefrontal cortex. Brain Res Bull 25:975–979
Imperato A, Mulas A, Di Chiara G (1986) Nicotine preferentially stimulates dopamine release in the limbic system of freely moving rats. Eur J Pharmacol 132:337–338
Izenwasser S, Cox BM (1992) Inhibition of dopamine uptake by cocaine and nicotine: tolerance to chronic treatments. Brain Res 573:119–125
Kalivas PW, Duffy P (1990) Effect of acute and daily cocaine treatment on extracellular dopamine in the nucleus accumbens. Synapse 5:48–58
Kita T, Okamoto M, Nakashima T (1992) Nicotine-induced sensitization to ambulatory stimulant effect produced by daily administration into the ventral tegmental area and the nucleus accumbens in rats. Life Sci 50:583–590
Ksir C, Hakan RL, Kellar KJ (1987) Chronic nicotine and locomotor activity: influences of exposure dose and test dose. Psychopharmacology 92:25–29
Johnson DH, Engel JA, Soderpalm B (1995) A time-course analysis of behavioral sensitization to nicotine in the rat. Neurosci Abstr 574.18
Lapin EP, Maker HS, Sershen H, Hurd Y, Lajtha A (1987) Dopamine-like action of nicotine: lack of tolerance and reverse tolerance. Brain Res 407:351–363
Mark GP, Schwartz DH, Hernandez L, West HL, Hoebel BG (1991) Application of microdialysis to the study of motivation and conditioning: measurements of dopamine and serotonin in freely behaving rats. In: Robinson TE, Justice JB (eds) Microdialysis and the neurosciences. Elsevier, New York, pp 369–385
MacLennan AJ, Maier SF (1983) Coping and stress induced potentiation of stimulant stereotypy in the rat. Science 219:1091–1093
Mitchell SN, Brazell MP, Joseph MH, Alavijeh MS, Gray JA (1989) Regionally specific effects of acute and chronic nicotine on rates of catecholamine and 5-hydroxytryptamine synthesis in rat brain. Eur J Pharmacol 167:311–322
Museo I, Wise RA (1990a) Locomotion induced by ventral tegmental microinjections of a nicotinic agonist. Pharmacol Biochem Behav 35:735–737
Museo I, Wise RA (1990b) Microinjections of a nicotinic agonist into dopamine terminal fields: effects on locomotion. Pharmacol Biochem Behav 37:113–116
Paxinos G, Watson C (1986) The rat brain in stereotaxic coordinates. Academic Press, San Diego
Pert A, Post RM, Weiss SRB (1990) Conditioning as a critical determinant of sensitization produced by psychostimulants. In: Erinoff L (ed) NIDA Research Monograph 97, US Government Printing, Washington DC, pp 208–241
Post RM, Weiss SRB, Fontana D, Pert A (1992) Conditioned sensitization to the psychomotor stimulant cocaine. In: The neurobiology of drug and alcohol addiction. Ann NY Acad Sci 654:386–399
Reid MS, Hsu K, Tolliver BK, Berger SP (1995) Evidence for the involvement of phospholipase A2 mechanisms in the development of stimulant sensitization. J Pharmacol Exp Ther 276:1244–1256
Schenk S, Snow S, Horger BA (1991) Pre-exposure to amphetamine but not nicotine sensitizes rats to the motor activating effect of cocaine. Psychopharmacology 103:62–66
Schiff SR (1982) Conditioned dopaminergic activity. Biol Psychiatry 17:135–154
Shoaib M, Stolerman IP (1992) MK801 attenuates behavioral adaptation to chronic nicotine administration. Br J Pharmacol 105:514–515
Shoaib M, Benwell MEM, Akbar MT, Stolerman IP, Balfour DJK (1994) Behavioral and neurochemical adaptations to nicotine in rats: influence of NMDA antagonists. Br J Pharmacol 111:1073–1080
Stewart J, de Wit H, Eikelboom R (1984) Role of unconditioned and conditioned drug effects in the self-administration of opiates and stimulants. Psychol Rev 91:251–268
Stolerman IP, Reavill C (1989) Primary cholinergic and indirect dopaminergic mediation of behavioral effects of nicotine. Prog Brain Res 79:227–237
Suemara K, Gomita Y, Furuno K, Araki Y (1993) Chronic nicotine treatment potentiates behavioral responses to dopaminergic drugs in rats. Pharmacol Biochem Behav 46:135–139
Tolliver BK, Belknap JK, Woods WE, Carney JM (1994) Genetic analysis of sensitization and tolerance to cocaine. J Pharmacol Exp Ther 270:1230–1238
Vezina P, Blanc G, Glowinski J, Tassin J-P (1992) Nicotine and morphine differentially activate brain dopamine in prefrontal and subcortical terminal fields: effects of acute and repeated injections. J Pharmacol Exp Ther 261:484–490
Vezina P, Herve D, Glowinski J, Tassin JP (1994) Injections of 6-hydroxydopamine into the ventral tegmental area destroy mesolimbic dopamine neurons but spare the locomotor activating effects of nicotine in the rat. Neurosci Lett 186:111–114
Walter S, Kuschinsky K (1989) Conditioning of nicotine effects on motility and behavior in rats. Naunyn-Schmiedeberg's Arch Pharmacol 339:208–213
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Reid, M.S., Ho, L.B. & Berger, S.P. Effects of environmental conditioning on the development of nicotine sensitization: behavioral and neurochemical analysis. Psychopharmacology 126, 301–310 (1996). https://doi.org/10.1007/BF02247381
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02247381