Abstract
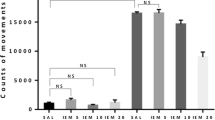
The influence of cholecystokinin (CCK), bilaterally injected into the rostral nucleus accumbens, on the EEG and behavioural effects induced by phencyclidine (PCP) has been studied in rats. CCK (10 ng) significantly inhibited PCP-induced EEG effects (increase of spectral power with respect to pre-drug tracing; increase of relative power distribution in the slowest frequency bands), and behavioural effects (circling and ataxia). The inhibitory effects of CCK were completely antagonized by 1 ng PD 135–158, a selective CCKB receptor antagonist, but not by lorglumide (1 µg), a selective CCKA receptor antagonist. Since the effects induced by PCP in rodents have been proposed to be an experimental correlate of the psychotic symptoms it induces in humans, these results indicate that CCK may act as a neuroleptic. They also suggest that CCKB receptors located in the rostral nucleus accumbens may be involved in the neuroleptic-like activity of CCK.
Similar content being viewed by others
References
Altar CA, Boyar WC (1989) Brain CCK-B receptors mediate the suppression of dopamine release by cholecystokinin. Brain Res 483:321–326
Anis NA, Berry SC, Burton NR, Lodge D (1983) The dissociative anaesthetics, ketamine and phencyclidine selectively reduce excitation of mammalian neurones byN-methyl-aspartate. Br J Pharmacol 79:565–575
Bagchi SP (1981) Effects of phencylidine on synaptosomal dopamine continuously appearing from phenylalanine: sensitivity to reserpine. Neuropharmacology 20:845–851
Bakker CB, Amini FB (1961) Observation on the psychotomimetic effects of sernyl. Comp Psychiatry 2:269–273
Castellani S, Adams PM (1981) Acute and chronic phencyclidine effects on locomotor activity, stereotypy and ataxia in rats. Eur J Pharmacol 73:143–154
Crawley JN, Stivers JA, Blumstein LK, Paul SM (1985) Cholecystokinin potentiates dopamine-mediated behaviours: evidence for modulation specific to a site of coexistence. J Neurosci 5:1972–1983
Crawley JN, Stivers JA, Hommer DW, Skirboll LR, Paul SM (1986) Antagonists of central and peripheral behavioural actions of cholecystokinin. J Pharmacol Exp Ther 236:320–330
De Witte P, Heidbreder C, Roquest B, Vanderhaeghen JJ (1987) Opposite effects of cholecystokinin octapeptide (CCK-8) and tetrapeptide (CCK-4) after injection into the caudal part of the nucleus accumbens or into its rostral part and the cerebral ventricles. Neurochem Int 10:473–479
Domino EF (1964) Neurobiology of phencyclidine (Sernyl), a drug with an unusual spectrum of pharmacological activity. Int Rev Neurobiol 6:303–347
Fauman BJ, Aldinger G, Fauman M, Rosen P (1976) Psychiatric sequelae of phencyclidine abuse. Clin Toxicol 9:529–538
Freeman AS, Bunney BS (1984) The effects of phencyclidine andN-allylnormetazocine on midbrain dopamine neuronal activity. Eur J Pharmacol 104:287
Fuxe K, Agnati LF, Celani MF (1983) Evidence for interactions between striatal cholecystokinin and glutamate receptors. CCK-8 in vitro produces a marked downregulation of3H-glutamate binding sites in striatal membranes. Acta Physiol Scand 118:75–77
Garey RE, Heath RG (1976) Effects of phencyclidine on the uptake of H-catecholamines by rat striatal and hypothalamic synaptosomes. Life Sci 18:1105–1110
Hokfelt T, Rehfeld JF, Skirboll L, Ivemark B, Goldstein M, Markey K (1990) Evidence for coexistence of dopamine and CCK in mesolimbic neurones. Nature 285:476–478
Hughes J, Boden P, Costall B, Domeney A, Kelly E, Horwell DC, Hunter JC, Pinnock RD, Woodruff GN (1990) Development of a class of selective cholecystokinin type B receptor antagonists having potent anxiolytic activity. Proc Natl Acad Sci USA 87:6728–6732
Innis RB, Snyder SH (1980) Distinct cholecystokinin receptors in brain and pancreas. Proc Natl Sci USA 77:6917–6921
Itoh S, Lal H (1990) Influences of cholecystokinin and analogues on memory processes. Drug Dev Res 21:257–276
Lindefors N, Lindén A, Brené S, Sedvall G, Persson H (1993) CCK peptides and mRNA in the human brain. Prog Neurobiol. 40:671–690
Luby ED, Gottlieb JS, Cohen BD, Rosenbaum G, Domino EF (1962) Model psychoses and schizophrenia. Am J Psychiatry 119:61–67
Makovec F, Bani M, Cereda R, Chisté R, Pacini MA, Revel L, Rovati A, Rovati LC, Setnikar I (1987) Pharmacological properties of lorglumide as a member of a new class of cholecystokinin antagonists. Arzneimittelforschung Drug Res 37:1265–1268
Marshall FH, Barnes S, Hughes J, Woodruff GN, Hunter JC (1991) Cholecystokinin modulates the release of dopamine from the anterior and posterior nucleus accumbens by two different mechanisms. J Neurochem 56:917–922
Meltzer HY, Stahl SM (1976) The dopamine hypothesis of schizophrenia. Schizophr Bull 2:19–76
Montgomery SA, Green MCD (1988) The use of cholecystokinin in schizophrenia: a review. Psychol Med 18:593–603
Mutt V, Jorpes JE (1968) Structure of porcine cholecystokinin-pancreozymin. 1. Clearage with thrombin and trypsin. Eur J Biochem 6:156–162
Nair NPV, Lal S, Bloom DM (1986) Cholecystokinin and schizophrenia. Prog Brain Res 65:237–258
Popoli P, Caporali MG, Scotti de Carolis A (1990a) Evidence for the involvement of D-1 dopamine receptors in PCP-induced stereotypy and ataxia in rabbits. Pharmacol Res 22:197–205
Popoli P, Benedetti M, Scotti de Carolis A (1990b) Influence of nimodipine and diltiazem, alone and in combination, on phencyclidine-induced effects in rats: an EEG and behavioural study. Eur J Pharmacol 191:141–148
Popoli P, Pèzzola A, Sagratella S (1994) Diphenylydantoin potentiates the EEG and behavioural effects induced byN-methyl-d-aspartate antagonists in rats. Psychopharmacology 113:471–475
Sagratella S, Benedetti M, Pèzzola A, Scotti de Carolis A (1989) Behavioural and electroencephalographic effects of excitatory amino acid antagonists and sigma opiate/phencyclidine-like compounds in rats. Neuropharmacology 28:57–61
Sagratella S, Scotti de Carolis A, Pèzzola A, Popoli P (1991) Behavioural and electroencephalographic interactions between haloperidol and PCP/sigma ligands in the rat. Psychopharmacology 105:485–491
Sherman AD, Hegwood TS, Baruah S (1991) Deficient NMDA-mediated glutamate release from synaptosomes of schizophrenics. Biol Psychiatry 30:1191–1198
Snell LD, Johnson KM (1985) Antagonism ofN-methyl-d-aspartate-induced transmitter release in the rat striatum by phencyclidine-like drugs and its relationship to turning behaviour. J Pharmacol Exp Ther 235:50–57
Studler JM, Reibaud M, Herve D, Blanc G, Glowinski J, Tassin JP (1986) Opposite effects of sulphated cholecystokinin on DA-sensitive adenylate cyclase in two areas of the rat nucleus accumbens. Eur J Pharmacol 126:125–128
Vaccarino FJ, Rankin J (1989) Nucleus accumbens cholecystokinin (CCK) can either attenuate or potentiate amphetamine-induced locomotor activity: evidence for rostral-caudal differences in accumbens CCK function. Behav Neurosci 103:831–836
Vanderhaeghen JJ, Signeau JC, Gepts W (1975) New peptide in the vertebrate CNS reacting with antigastrin antibodies. Nature 257:604–605
Vickroy TW, Johnson KM (1981) Stimulation of synaptosomal tyrosine hydroxylation by phencyclidine in vitro. Eur J Pharmacol 71:463–473
Vickroy TW, Johnson KM (1982) Similar dopamine-releasing effects of phencyclidine and nonamphetamine stimulants in striatal slices. J Pharmacol Exp Ther 223:669–674
Vickroy TW, Bianchi RB, Kerwin JF, Kopecka H, Nadzan AM (1988) Evidence that type A CCK receptors facilitate dopamine efflux in rat brain. Eur J Pharmacol 152:371–372
Willetts J, Balster RL, Leander JD (1990) The behavioural pharmacology of NMDA receptor antagonists. Trends Pharmacol Sci 11:423–428
Winsauer PJ, Thompson DM (1991) Differential interaction of cholecystokinin with morphine and phencyclidine: effects on operant behavior in pigeons. Pharmacol Biochem Behav 41:83–90
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Popoli, P., Reggio, R., Pèzzola, A. et al. The stimulation of cholecystokinin receptors in the rostral nucleus accumbens significantly antagonizes the EEG and behavioural effects induced by phencyclidine in rats. Psychopharmacology 120, 156–161 (1995). https://doi.org/10.1007/BF02246188
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02246188