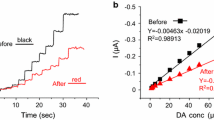
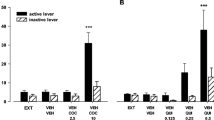
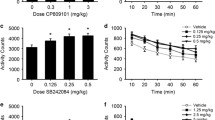
Abstract
The influence of the CCK-A antagonist devazepide and the CCK-B/gastrin antagonist L-365,260 on the locomotor activity of mice was studied. Devazepide and L-365,260 had opposite effects on spontaneous locomotor activity, and on caerulein- and apomorphine-induced hypomotility in the mouse. Devazepide in high doses (0.1–1 mg/kg IP) reduced spontaneous motor activity, whereas L-365,260 at a high dose (1 mg/kg IP) increased the activity of mice. Devazepide (0.1–10 µg/kg) moderately antagonized the sedative effect of apomorphine (0.1 mg/kg SC) and caerulein (25 µg/kg SC), whereas L-365,260 (1–10 µg/kg) significantly potentiated the actions of dopamine and CCK agonists. Concomitant administration of caerulein (15 µg/kg SC) and apomorphine (0.1 mg/kg SC) caused an almost complete loss of locomotor activity in the mouse. Devazepide and L-365,260 (0.1–10 µg/kg) were completely ineffective against caerulein-induced potentiation of apomorphine hypomotility. Devazepide in high doses (0.1–1 mg/kg), reducing the spontaneous motor activity of mice, counteracted the motor excitation induced byd-amphetamine (5 mg/kg IP). The CCK agonist caerulein (100 µg/kg SC) had a similar antiamphetamine effect. Devazepide (1–100 µg/kg) and L-365,260 (1 µg/kg) reversed completely the antiamphetamine effect of caerulein. The results of present study reflect apparently distinct role of CCK-A and CCK-B receptors in the regulation of motor activity. The opposite effect of devazepide and L-365,260 on caerulein- and apomorphine-induced hypolocomotion is probably related to the antagonistic role of CCK-A and CCK-B receptor subtypes in the regulation of mesencephalic dopaminergic neurons. The antiamphetamine effect of caerulein is possibly linked to the stimulation of CCK-A receptors in the mouse brain, whereas the blockade of both subtypes of the CCK-8 receptor is involved in the antiamphetamine effect of devazepide.
Similar content being viewed by others
References
Altar CA, Boyar WC (1989) Brain CCK-B receptors mediate the suppression of dopamine release by cholecystokinin. Brain Res 483:321–326
Barrett RW, Steffey ME, Wolfram CAW (1989) Type-A CCK sites in cow brain: characterization using (−)-[3H]-L-364,718 membrane binding assays. Mol Pharmacol 36:285–290
Bradbury AJ, Costall B, Naylor RJ, Neumeyer JL (1983) Motor inhibition induced by apomorphine derivatives in the mouse. J Pharm Pharmacol 35:494–499
Chang RSL, Lotti VJ (1986) Behavioral and pharmacological characterization of an extremely potent and selective nonpeptide cholecystokinin antagonist. Proc Natl Acad Sci USA 83:4923–4926
Costall B, Domeney AM, Naylor RJ (1985) The continuity of dopamine receptors antagonism can dictate the long-term behavioural consequences of a mesolimbic infusion of dopamine. Neuropharmacology 24:193–197
Crawley JN (1989) Microinjection of cholecystokinin into the rat ventral tegmental area potentiates dopamine-induced hypolocomotion. Synapse 3:346–355
Crawley JN, Kiss JZ (1985) Tracing the sensory pathway from the gut to brain regions mediating the actions of cholecystokinin on feeding and exploration. Ann NY Acad Sci 448:586–588
Crawley JN, Schwaber JS (1984) Abolition of the behavioral effects of cholecystokinin following bilateral radiofrequency lesions of parvocellular subdivision of the nucleus tractus solitarius. Brain Res 295:289–299
Crawley JN, Stivers JA, Blumstein LK, Paul SM (1985) Cholecystokinin potentiates dopamine-mediated behaviors: evidence for modulation specific to a site of coexistence. J Neurosci 5:1972–1983
De Witte P, Goldman S, Gewiss M, Poels JF, Van Boxez P, Van Der Veken E, Vanderhaeghen JJ (1986) Similar effect of caerulein on intracranial self-stimulation in vagotomized and non-vagotomized rats. Neurochem Int 8:339–343
Dourish CT, Hill DR (1987) Classification and function of CCK receptors. TIPS 8:207–208
Dourish CT, Rycroft N, Iversen SD (1989) Postponement of satiety by blockade of brain cholecystokinin (CCK-B) receptors. Science 245:1509–1511
Dourish CT, O'Neill MF, Coughlan J, Kitchener SJ, Hawley D, Iversen SD (1990) The selective CCK-B antagonist L-365,260 enhances morphine analgesia and prevents morphine tolerance in the rat: evidence for mediation of CCK/opioid interactions by CCK-B receptor. Eur J Pharmacol 176:35–44
Gerhardt GA, Friedmann M, Brodie MS, Vickroy TW, Gratton AP, Hoffer BJ, Rose GM (1989) The effect of cholecystokinin (CCK-8) on dopamine-containing nerve terminals in the caudate nucleus and nucleus accumbens of the anaesthetized rat: an in vivo electrochemical studies. Brain Res 499:157–163
Hagino Y, Moroji T, Iizuka R (1989) A behavioural pharmacological study on intracerebroventricularly administered CCK-8 related peptides in mice. Neuropeptides 13:107–113
Hamamura T, Kazahaya Y, Otsuki S (1989) Ceruletide suppresses endogenous dopamine release via vagal afferent system studied by in vivo intracerebral dialysis. Brain Res 483:78–83
Hamilton MH, Rose IC, Herberg LJ, De Belleroche JS (1990) Effect of intracerebroventricular and systemic injections of caerulein, a CCK analogue, on electrical self-stimulation and its interaction with the CCK-A receptor antagonist, L-364,718 (MK-329). Psychopharmacology 101:384–389
Hill DR, Campbell NJ, Shaw TM, Woodruff GN (1987) Autoradiographic localization and characterization of peripheral type CCK receptors in rat CNS using highly selective nonpeptide CCK antagonists. J Neurosci 7:2967–2976
Hökfelt T, Rehfeld JF, Skirboll L, Ivemark B, Goldstein M, Markey K (1980) Evidence for coexistence of dopamine and CCK in mesolimbic neurones. Nature 285:476–478
Hommer DW, Palkovits M, Crawley JN, Paul SM, Skirboll LR (1985) Cholecystokinin-induced excitation in the substantia nigra: evidence for peripheral and central components. J Neurosci 5:1387–1397
Hommer DW, Stoner G, Crawley JN, Paul SM, Skirboll LR (1986) Cholecystokinin-dopamine coexistence: electrophysiological actions corresponding to cholecystokinin receptor subtype. J Neurosci 6:3039–3043
Khosla S, Crawley JN (1988) Potency of L-364,718 as an antagonist of the behavioural effects of peripherally administered cholecystokinin. Life Sci 42:153–159
Lotti VJ, Chang RSL (1989) A new potent and selective non-peptide gastrin antagonist and brain cholecystokinin receptor (CCK-B) ligand: L-365,260. Eur J Pharmacol 162:273–280
Matsubara K, Matsushita A (1986) β-endorphin involvement in the antidopaminergic effect of caerulein. Jpn J Pharmacol 40:417–422
Moran TH, Robinson PH, Goldrich M, Mc Hugh PR (1986) Two brain CCK receptors: implications for behavioral actions. Brain Res 362:175–179
Morley JE (1987) Behavioural effects of administered cholecystokinin. ISI Atlas Pharmacol 1:49–51
Moroji T, Hagino Y (1987) Bilateral subdiaphragmatic vagotomy does not prevent the behavioural effects of systematically administered ceruletide in mice. Neuropeptides 9:217–224
Rovati L (1988) New pentanoic acid derivatives with potent CCK antagonistic properties: different activity on periphery vs. central nervous system. In: Wang RY, Schoenfeld R (eds) Cholecystokinin antagonists. Liss, New York, pp 1–11
Soar J, Hewson G, Leighton GE, Hill RG, Hughes J (1989) L-364,718 antagonizes the cholecystokinin-induced suppression of locomotor activity. Pharmacol Biochem Behav 33:637–640
Strömbom U (1977) Antagonism by haloperidol of locomotor depression induced by small doses of apomorphine. J Neural Transm 40:191–194
Vasar E, Allikmets L, Ryzhov I, Prakhye I, Soosaar A, Mirzayev S (1988) Intraspecies differences in the behavioural effects of caerulein, an agonist of CCK-8 receptors, in mice and rats. Bull Exp Biol Med 105:168–171
Vickroy TW, Bianchi BR (1989) Pharmacological and mechanistic studies of cholecystokinin-facilitated [3H]-dopamine efflux from rat nucleus accumbens. Neuropeptides 13:43–50
Zetler G (1985) Neuropharmacological profile of cholecystokinin-like peptides. Ann NY Acad Sci 448:448–469
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vasar, E., Harro, J., Lang, A. et al. Differential involvement of CCK-A and CCK-B receptors in the regulation of locomotor activity in the mouse. Psychopharmacology 105, 393–399 (1991). https://doi.org/10.1007/BF02244435
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02244435