Summary
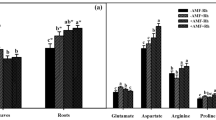
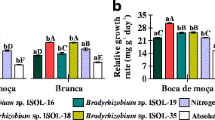
The effect of variousRhizobium trifolii strains on the nitrogenase activity profiles of red clover (Trifolium pratense cv. Venla) was studied in greenhouse and field experiments. The nitrogenase activity of nodulated roots or whole plants was measured with the acetylene reduction assay at different stages of plant growth. The mean nitrogenase activity followed plant shoot growth rate from the time when activity was first detected to the onset of flowering. There was a positive correlation between the nitrogenase activity at certain stages of growth and the final harvest yields, whereas at other stages the correlation was absent or negative. Acetylene reduction activity measured at the early stages of plant growth, when the correlation was positive, could be used to screen for high-yielding symbioses betweenR. trifolii and the red clover cultivar Venla. The results suggest that the high-yielding symbioses could be characterized by a nitrogenase activity profile closely linked to plant growth rate, and by a comparatively high nitrogenase activity at early stages of development, the lower-yielding symbioses showing larger deviations of nitrogenase activity from the mean relative shoot growth rate, and being less active at early growth stages.
Similar content being viewed by others
References
Ball R, Brougham R W, Brock J L, Crush J R, Hoglund J H and Carran R A 1979 Nitrogen fixation in pasture. I. Introduction and general methods. N. Z. J. Exp. Agric. 7, 1–5.
Bethlenfalvay G J, Abu-Shakra S S and Phillips D A 1978 Interdependence of nitrogen nutrition and photosynthesis inPisum sativum L. I. Effect of combined nitrogen on symbiotic nitrogen fixation and photosynthesis. Plant Physiol. 62, 127–130.
Bethlenfalvay G J, Abu-Shakra S S and Phillips D A 1978 Interdependence of nitrogen nutrition and photosynthesis inPisum sativum L. II. Host plant response to nitrogen fixation by Rhizobium strains. Plant Physiol. 62, 131–133.
Duhigg P, Melton B and Baltensperger A 1978 Selection for acetylene reduction rates in ‘Mesilla’ alfalfa. Crop Sci. 18, 813–816.
Farrington P E, Greenwood E A N, Titmanis Z V, Trinick M J and Smith D W 1977 Fixation, accumulation and distribution of nitrogen in a crop ofLupinus angustifolius cv. Unicrop. Aust. J. Agric. Res. 28, 237–248.
Gibson A H 1967 Carbon dioxide limitations of plant growth in tube culture with special reference to legume nodulation studies. Aust. J. Biol. Sci. 20, 837–842.
Goh K M, Edmeades D C and Robinson B W 1978 Field measurements of symbiotic nitrogen fixation in an established pasture using acetylene reduction and a15N method. Soil Biol. Biochem. 10, 13–20.
Halliday J, and Pate J S 1976 The acetylene reduction assay as a means of studying nitrogen fixation in white clover under sward and laboratory conditions. J. Brit. Grassl. Soc. 31, 29–35.
Ham G E, Lawn R J and Brun W A 1976 Influence of inoculation, nitrogen fertilizers and photosynthetic source-sink manipulations on field-grown soybeans.In Symbiotic Nitrogen Fixation in Plants. Ed. PS Nutman. Cambridge University Press, London, pp 239–253.
Hardy R W F, Burns R C, Herbert R R, Holsten R D and Jackson E K 1971 Biological nitrogen fixation: a key to world protein. Plant and Soil, Spec. Vol. pp 561–590.
Hardy R W F and Havelka U D 1976 Photosynthate as a major factor limiting nitrogen fixation by field-grown legumes with emphasis on soybeans.In Symbiotic Nitrogen Fixation in Plants. Ed. P S Nutman. Cambridge University Press, London, pp 421–439.
Lindström K 1984 Analysis of factors affectingin situ nitrogenase activity ofGalega orientalis, Trifolium pratense andMedicago sativa in temperate conditions. Plant and Soil 79,In press.
Masterson C L and Murphy P M 1976 Application of the acetylene reduction technique to the study of nitrogen fixation by white clover in the field.In Symbiotic Nitrogen Fixation in Plants. Ed. P S Nutman. Cambridge University Press, London, pp 299–316.
Minchin F R, Witty J F, Sheehy J E and Müller M 1983 A major error of the acetylene reduction assay: decrease in nodular nitrogenase activity under assay conditions. J. Exp. Bot. 34, 641–649.
Pate J S, Atkins C A and Rainbird R M 1981 Theoretical and experimental costing of nitrogen fixation and related processes in nodules of legumes.In Current Perspectives in Nitrogen Fixation. Eds. A H Gibson and W E Newton. Australian Academy of Science, Canberra, pp 105–116.
Phillips D A and Bennett J P 1978 Measuring symbiotic nitrogen fixation in rangeland plots ofTrifolium subterraneum L. andBromus mollis L. Agron. J. 70, 671–674.
Rice W A 1980 Seasonal patterns of nitrogen fixation and dry matter production by clovers grown in the Peace River region. Can. J. Plant Sci. 60, 847–858.
Schubert K R and Evans H J 1976 Hydrogen evolution: A major factor affecting the efficiency of nitrogen fixation in nodulated symbionts. Proc. Nat. Acad. Sci. USA 73, 1207–1211.
Sinclair A G, Hannagan R and Risk W H 1976 Evaluation of the acetylene reduction assay of nitrogen fixation using small soil-core samples. N.Z. J. Agric. Res. 19, 451–458.
Sprent J J and Bradford A M 1977 Nitrogen fixation in field beans (Vicia faba) as affected by population density, shading and its relationship with soil moisture. J. Agric. Sci. (Camb.) 88, 303–310.
Sprent J J, Bradford A M and Gallacher A E 1978 Factors affecting nodulation and nitrogen fixation byVicia faba. Ann. Appl. Biol. 88 473–476.
Vincent J M 1970 A Manual for the Study of Root-Nodule Bacteria. IBP Handbook 15, Blackwell Scientific Publications, Oxford and Edinburgh.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lindström, K. Effect of variousRhizobium trifolii strains on nitrogenase (C2H2) activity profiles of red clover (Trifolium pratense cv. Venla). Plant Soil 80, 79–89 (1984). https://doi.org/10.1007/BF02232941
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02232941