Abstract
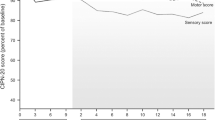
38 patients with glial brain tumors received 135 mg/m2 cisplatin intravenously every month for 5 courses. Signs and symptoms of peripheral neuropathy were evaluated clinically and electrophysiologically. This approach differs from methods previously reported in that it offers two major advantages: primary brain tumors do not cause paraneoplastic neuropathy; no neurotoxic drugs other than cisplatin were employed. Our study confirmed in this highly specific clinical model the existence of cisplatin peripheral neuropathy: we observed definite clinical signs or symptoms of neuropathy in 80% of patients receiving a cumulative cisplatin dosage of 675 mg/m2; there was a progressive dose-related decrease in SAP amplitudes, expression of a sensory axonopathy or neuronopathy.
Sommario
Trentotto soggetti portatori di tumori cerebrali della serie gliale sono stati trattati con 5 cicli mensili di cisplatino, ciascuno di 135 mg/m2, per via endovenosa. Segni e sintomi di neuropatia periferica sono stati valutati clinicamente ed elettrofisiologicamente. Il nostro approccio differisce dai precedenti lavori riportati in letteratura in quanto offre due importanti vantaggi: i tumori cerebrali primitivi non causano mai neuropatie paraneoplastiche; non è stato usato nessun altro farmaco neurotossico all'infuori del cisplatino. Lo studio conferma l'esistenza della neuropatia periferica indotta dal cisplatino anche in questo modello altamente specifico. Abbiamo potuto osservare chiari segni clinici di neuropatia periferica nell'80% dei pazienti trattati con una dose cumulativa di cisplatino di 675 mg/m2. Dal punto di vista eletrofisiologico si è osservata una riduzione progressiva e dose-correlata nell'ampiezza dei SAP, espressione di una neuropatia sensitiva assonale e/o neuronale.
Similar content being viewed by others
References
Ashraf M., Scotchel P.L., Krall J.M., Fink E.B.:Cis-Platinum-induced hypomagnesemia and peripheral neuropathy. Gynecol. Oncol. 16:309–318, 1983.
Cavaletti G., Tredici G., Pizzini G., Minoia A.:Tissue platinum concentrations and cisplatin schedules. Lancet 336:1003, 1990.
Gerritsen van der Hoop R., Vecht C.J., van der Burg M.e.l., et al.:Prevention of cisplatin neurotoxicity with an ACTH (4-9) analogue in patients with ovarian cancer. N. Engl. J. Med. 322:89–94, 1990.
Karnofsky D.A., Burchenal J.H.:The clinical evaluation of chemoterapeutic agents in cancer. In: Evaluation of chemoterapeutic agents, McLeod C.M., ed., New York: Columbia University Press, 191–205, 1949.
Khan A.B., D'Souza B.J., Wharam M., et al.:Cisplatin in therapy in recurrent childhood brain tumors. Cancer Treat. Rep. 66:2013, 1982.
Kimura J.:Principles of nerve conduction studies.pp. In: Electrodiagnosis in diseases of nerve and muscle: principles and practice. Philadelphia: F.A. Davis Company, Ed 2, 78–102, 1989.
Mollman, J.E.:Cisplatin neurotoxicity. N. Engl. J. Med. 322:126–127, 1990.
Roelofs R.I., Hrushesky W., Rogin J., Rosenberg L.:Peripheral sensory neuropathy and cisplatin chemotherapy. Neurology 34:934–938, 1984.
Steward D.J., Leavens M., Feun M., et al.:Human central nervous system distribution of cisplatin and use as radiosensitizer in malignant brain tumors. Cumuliforme-results of phase II study. Int. J. Radiat. Oncol. Biol. Phys. 8:1787–1794, 1982.
Thompson S.W., Davis L.E., Kornfeld M., Hilgers R.D., Standefer J.C.:Cisplatin neuropathy. clinical, electrophysiologic, morphologic, and toxicologic studies. Cancer 54:1269–75, 1984.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sghirlanzoni, A., Silvani, A., Scaioli, V. et al. Cisplatin neuropathy in brain tumor chemotherapy. Ital J Neuro Sci 13, 311–315 (1992). https://doi.org/10.1007/BF02223095
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02223095