Abstract
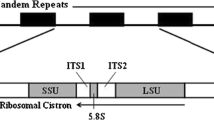
The non-transcribed spacer of the ribosomal DNA repeat ofPythium pachycaule was found to exist in two versions, one about 200 by longer than the other. Both versions contained a conserved 5s rRNA gene in inverted orientation and a diverged copy of the 5s sequence as a tandem repeat, with a spacer of about 180 by between them. This is the first report of multiple 5s-like sequences in the non-transcribed spacer of the rDNA of any organism. The two diverged 5s sequences differ from each other at nine positions in the 5′ half of the sequence, but they have the same central 10-bp deletion and their 3′ halves are identical. The diverged sequences, 5s and 5s″, when aligned with the 5s sequence, have the same base at 63.9 and 58.8% of the positions respectively. The spacers between 5s sequences in both versions were highly conserved. The secondary structures of potential rRNA products of the diverged 5s sequences indicate that they may be pseudogenes. The relationships between the functional 5s gene and the diverged gene copies suggest that the 5s sequence family ofP. pachycaule originated by duplication of an ancestral gene, followed by divergence and the introduction of heterogeneity. This work shows the potential for tandemization of 5s genes in the NTS, and may shed light on the evolutionary events that led to the transition from the rDNA-linked 5s genes found inPythium species with filamentous zoosporangia to the unlinked tandem arrays found inPythium species with globose zoosporangia.
Similar content being viewed by others
References
Ali-Shtayeh MS, Dick MW (1985) Five new species ofPythium (Peronosporomycetidae). Bot J Linnean Soc 91:297–317
Bartnik E, Bartoszeweski S, Borsuk P, Empel J (1986)Aspergillus nidulans 5s rRNA genes and pseudogenes. Curr Genet 10:453–457
Belkhiri A, Buchko J, Klassen G (1992) The 5s ribosomal RNA gene inPythium species: two different genomic locations. Mol Biol Evol 9:1089–1102
Buchko J, Klassen G (1990) Detection of length heterogeneity in the ribosomal DNA ofPythium ultimum by PCR amplification of the intergenic region. Curr Genet 18:203–205
Dams E, Hendriks L, Van de Peer Y, Neefs J-M, Smits, Vandenbempt I, De Wachter R (1988) Compilation of small ribosomal subunit RNA sequences. Nucleic Acids Res 16:r87-r173
Dick MW (1990) Keys toPythium. MW Dick, Reading, UK
Francis DM, St. Clair DA (1993) Outcrossing in the homothallic oomycetePythium ultimum detected with molecular markers. Curr Genet 24:100–106
Free SJ, Rice PW, Metzenberg RL (1979) Arrangement of the genes coding for ribosomal ribonucleic acids inNeurospora crassa. J Bacteriol 137:1219–1226
Gutell RR, Fox GE (1988) A compilation of large subunit RNA sequences presented in a structural format. Nucleic Acids Res 16:rl75-r269
Hausner G, Eyolfsdottir GG, Reid J, Klassen GR (1992) Two additional species of the genusTogninia. Can J Bot 70:124–134
Howlet BJ, Brownlee AG, Guest DI, Adcock GJ, McFadden GI (1992) The 5s ribosomal RNA gene is linked to the large and small subunit ribosomal RNA genes in the oomycetesPhytophthora vignae, P. cinnamomi, P. megasperma f.sp.glycinea andSaprolegnia ferax. Curr Genet 22:445–461
Jacq C, Miller JR, Brownlee GG (1977) A pseudogene structure in 5s DNA ofXenopus laevis. Cell 12:109–120
Kay BK, Gall JG (1981) 5s ribosomal RNA genes of the newtNotophthalmus viridescens. Nucleic Acids Res 9:6457–6469
Kim WK, Mauthe W, Hausner G, Klassen GR (1990) Isolation of high-molecular-weight DNA and double-stranded RNAs from fungi. Can J Bot 68:1898–1902
Klassen GR, Buchko J (1990) Subrepeat structure of the intergenic region in the ribosomal DNA of the oomycetous fungusPythium ultimum. Curr Genet 17:125–127
Maniatis T, Fritsch EF, Sambrook J (1982) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, New York
Martin F (1990) Variation in the ribosomal DNA repeat unit within single-oospore isolates of the genusPythium. Genome 33:585–591
Pieler T, Theunissen O (1993) TFIIIA: nine fingers-three hands? Trends Biochem Sci 18:226–230
Pieler T, Hamm J, Roeder RG (1987) The 5s gene internal control region is composed of three distinct sequence elements, organized as two functional domains with variable spacing. Cell 48:91–100
Rubacha A, Sumner III W, Richter L, Beckingham K (1984) Conserved 5′ flank homologies in dipteran 5s RNA genes that would function on “A” form DNA. Nucleic Acids Res 12:8193–8207
Saiki RK, Gelfand DH, Stoffel S, Scharf SJ, Higuchi R, Horn T, Mullis KB, Erlich HA (1988) Primer-directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science 239:487–491
Selker EU, Free SJ, Metzenberg RL, Yanofsky C (1981) An isolated pseudogene related to the 5s RNA genes inNeurospora crassa. Nature 294:576–578
Singh K, Bhatia S, Lakshmikumaran M (1994) Novel variants of the 5s rRNA genes inEruca sativa. Genome 37:121–128
Smith GP (1974) Unequal crossover and the evolution of multigene families. Cold Spring Harbor Symp Quant Biol 38:507–513
Tyler BM (1987) Transcription ofNeurospora crassa 5s RRNA genes requires a TATA box and three internal elements. J Mol Biol 196:801–811
Vahidi H, Curran J, Nelson DW, Webster JM, McClure MA, Honda BM (1988) Unusual sequences, homologous to 5s RNA, in ribosomal DNA repeats of the nematodeMeloidogyne arenaria. J Mol Evol 27:222–227
Walker WF, Doolittle WF (1982) Nucleotide sequences of 5s ribosomal RNA from four oomycete and chytrid water molds. Nucleic Acids Res 10:5717–5721
Wolters J, Erdmann VA (1988) Compilation of 5s rRNA gene sequences. Nucleic Acids Res 16:r1-r70
Author information
Authors and Affiliations
Additional information
Communicated by B.G. Turgeon
Rights and permissions
About this article
Cite this article
Belkhiri, A., Klassen, G.R. Diverged 5s rRNA sequences adjacent to 5s rRNA genes in the rDNA ofPythium pachycaule . Curr Genet 29, 287–292 (1996). https://doi.org/10.1007/BF02221560
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02221560