Abstract
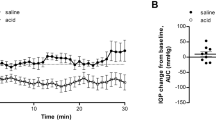
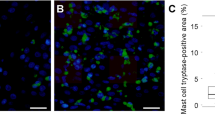
In previous studies we have demonstrated that microvascular permeability increases early in the course of experimental acid-induced esophageal mucosal injury. This is associated with an increase in the intraluminal appearance of histamine, suggesting a possible role for mast cells in this form of injury. In the present study, quantitative analysis of esophageal mast cells was undertaken using both light and electron microscopy in opossums undergoing intraluminal esophageal acid perfusion or normal saline control perfusion. Light microscopy showed that animals perfused with either 50 or 100 mM hydrochloric acid had an approximate 50% decrease in the number of stainable esophageal mast cells. Stereologic analysis of electron micrographs revealed that within the mucosa, the mean area of the mast cells as well as nuclear area and area of intact granules were also significantly reduced in acid perfused animals. Taken together these quantitative morphological analyses suggest that intraluminal acid exposure is associated with degranulation and/or lysis of esophageal mast cells and that released mediators from esophageal mast cells may play a role in the pathophysiology of reflux esophagitis.
Similar content being viewed by others
References
Ismail-Beigi F, Horton PF, Pope CE: Histological consequences of gastroesophageal reflux in man. Gastroenterology 58:163–174, 1970
Geboes K, Desmet V, Vantrappen G, Mebis J: Vascular changes in the esophageal mucosa. An early sign of esophagitis. Gastrointest Endosc 26:294–332, 1980
Collins BJ, Elliott H, Sloan JM, McFarland RJ, Love AHG: Oesophageal histology in reflux oesophagitis. J Clin Pathol 38:1265–1272, 1985
Zijlstra FG, Hynna-Liepert TT, Dinda PK, Beck IT, Paterson WG: Microvascular permeability increases early in the course of acid-induced esophageal injury. Gastroenterology 101:295–302, 1991
Barclay RL, Paterson WG, Dinda PK, Morris GP: Intraluminal acid perfusion induces histamine release from the opossum esophagus. Clin Invest Med 14(suppl A):A46, 1991 (abstract)
Theoharides TC: Histamine2 (H2)-receptor antagonists in the treatment of urticaria. Drugs 37:345–355, 1989
Casale TB, Wood D, Richerson HB, Zehr B, Zavala D, Hunninghake GW: Direct evidence of a role for mast cells in the pathogenesis of antigen-induced bronchoconstriction. J Clin Invest 80:1507–1511, 1987
Tiggelman AMBC, Van Noorden CJF: Mast cells in early stages of antigen-induced arthritis in rat knee joints. Int J Exp Pathol 71:455–464, 1990
Stanisz AM, Scicchitano R, Bienenstock J: Inflammatory Bowel Disease: Current Status and Future Approach. RP MacDermatt (ed.). Amsterdam, Elsevier, 1988, pp 49–54
Norris HT, Zamcheck N, Gottlieb LS. The presence and distribution of mast cells in the human gastrointestinal tract at autopsy. Gastroenterology 44:448–455, 1963
Strobel S, Miller HRP, Ferguson A: Human intestinal mucosal mast cells: evaluation of fixation and staining techniques. J Clin Pathol 34:851–858, 1981
Weibel ER: Stereological Methods, Vol I: Practical Methods for Biological Morphometry, London, Academic Press, 1979
Enerback L: Mast cells in rat gastrointestinal mucosa: 2. Dyebinding and metachromatic properties. Acta Pathol Microbiol Scand 66:303–312, 1966
DeMeester TR, Johnson LF, Joseph GJ, Toscano MS, Hall AW, Skinner DB: Patterns of gastroensophageal reflux in health and disease. Ann Surg 184:459–470, 1976
Stead RH, Dixon MF, Bramwell NH, Riddell RH, Bienenstock J: Mast cells are closely apposed to nerves in the human gastrointestinal mucosa. Gastroenterology 97:575–585, 1989
Newson B, Dahlstrom A, Enerback L, Ahlman H: Suggestive evidence for a direct innervation of mucosal mast cells: An electron microscopic study. Neuroscience 10:565–570, 1983
Perdue MH, Marshall J, Masson S: Ion transport abnormalities in inflamed rat jejunum: involvement of mast cells and nerves. Gastroenterology 98:561–567, 1990
Fewtrell CM, Foreman JC, Jordan CC, Oehme PL, Renner H, Stewart JM: The effects of substance P on histamine release and 5-hydroxytryptamine release in the rat. J Physiol (London) 330:393–411, 1982
Lippe IT, Holzer P: The rat gastric mucosal vasodilator respose to acid back diffusion is mediated by a neural reflex involving the celiac ganglion. Gastroenterology 100:A112, 1991 (abstract)
Berger W, Dahl G, Meissner HP: Structural and functional alterations in fused membranes of secretory granules during exocytosis in pancreatic islet cells of the mouse. Cytobiologic 12:119–135, 1975
Author information
Authors and Affiliations
Additional information
This work was supported by an operating grant from Physicians Services Incorporated of Ontario. Dr. Paterson is supported by an Ontario Ministry of Health Career Scientist Award.
Rights and permissions
About this article
Cite this article
Barclay, R.L., Dinda, P.K., Morris, G.P. et al. Morphological evidence of mast cell degranulation in an animal model of acid-induced esophageal mucosal injury. Digest Dis Sci 40, 1651–1658 (1995). https://doi.org/10.1007/BF02212685
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02212685