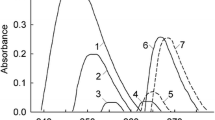
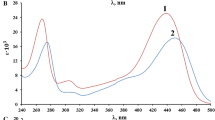
Abstract
The chemical behaviour of Cr/VI/ at low concentrations /10−6–10−7 mol.1−1/ in several aqueous formic acid solutions was studied using high specific activity51Cr/VI/. A simple and efficient radiochromatographic method using paired cation and anion exchange analyses was used to quantify the51Cr/VI/ remaining in solution after different time periods as well as the several51Cr/III/ species formed. The product distribution of monomeric51Cr/III/ species includes those with 0, +1, +2 and +3 charges. Very little dimeric or higher polymeric species are formed. The rate of reaction of51Cr/VI/ with formic acid depends on the acid concentration, the presence or absence of sodium chloride and wheter, or not, the samples are exposed to light.
Similar content being viewed by others
References
K. E. Collins, C. Archundia, C. H. Collins,Quím. Nova., 6 /1983/ 164.
C. Archundia, C. H. Collins, K. E. Collins, Proceeding of the Second General Congress on Nuclear Energy, Rio de Janeiro, April, 1988, Vol. 2, p. 433.
K. E. Collins, P. S. Bonato, C. Archundia, M. E. L. R. de Queiroz, C. H. Collins,Chromatographia, 26 /1988/ 160.
F. Hasan, J. Rocek,J. Am. Chem. Soc., 94 /1972/ 9073.
G. P. Haight, Jr., M. Rose, J. Preer,J. Am. Chem. Soc., 90 /1968/ 4809.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Archundia, C., Collins, C.H. & Collins, K.E. Speciation of the reaction products of51Cr/VI/ with formic acid. Journal of Radioanalytical and Nuclear Chemistry Letters 146, 49–56 (1990). https://doi.org/10.1007/BF02199271
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02199271